Introductory. The Elements Of The Science Of Nutrition. Part 3
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
Introductory. The Elements Of The Science Of Nutrition. Part 3
It has been stated that the form of Lavoisier's respiration apparatus is unknown. In 1850 Regnault and Reiset1 published an account of respiration experiments in which small animals were placed under a bell-jar containing a known quantity of oxygen. The air was kept free from carbon dioxid by pumping it through potassium hydrate, and oxygen was added from time to time. The gaseous exchange between the animal and its environment could be readily ascertained by determining the amount of carbon dioxid given off and the amount of oxygen absorbed. No attempt was made to determine from what materials the carbonic acid arose. The method of Regnault and Reiset placed the animals in a confined space where poisonous exhalations other than carbon dioxid could collect, and where the atmosphere became saturated with water. However, these factors were without influence on the health of their animals. They planned to work in one of the large hospitals in Paris, but, unfortunately, the project proved too costly and had to be renounced. They write, "L'etude de la respiration de l'homme dans ses divers etats pathologiques nous parait un des sujets les plus dignes d'occuper les hommes qui se vouent a l'art de guerir: elle peut donner un diagnostic precieux pour un grand nombre de maladies et rendre plus evidentes les revolutions qui survien-nent dans l'economie." Although Regnault and Reiset had no definite idea of the materials which were oxidized in the animals with which they were experimenting, we find that Bischoff and Voit1 tried to read such interpretations into the work of Regnault and Reiset. Thus Bischoff and Voit determined the quantity of nitrogen in the urine of a starving dog, which indicated that he had burned in twenty-four hours 218 grams of his own "flesh." The flesh was calculated from the nitrogen elimination on the basis of the knowledge that fresh meat contains 3.4 per cent, of nitrogen. Many of the older experiments were computed on this basis. It was shown that the 218 grams of "flesh" contained 40 grams of carbon. Bischoff and Voit estimate from the experiments of Regnault and Reiset that a meat-fed dog of a weight similar to the above would give off 250 grams of carbon and absorb 900 grams of oxygen in the respiration of twenty-four hours. These figures indicated to Bischoff and Voit that the extra carbon elimination was due to the combustion of fat, and they reached the conclusion that the waste of the body in starvation is dependent on the metabolism of protein and fat. Correct results, however, were attainable only by combining the two methods, so that both the quantity of the nitrogen and carbon of the urine and feces and the amount of carbon dioxid of the respiration during the same period of time could be ascertained. This was accomplished by the respiration apparatus of Pettenkofer.
1 Regnault and Reiset: "An. d. Chem. und Pharm.," 1850, lxxiii, 92,129, 257.
1 Bischoff and Voit: "Die Gesetze der Ernahrung des Fleischfressers," 1860, p. 43.
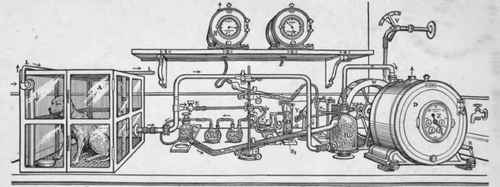
The smaller respiration apparatus of Pettenkofer and Voit. Air enters the cage, A, at the upper left-hand corner. It is drawn out through openings in the tube B (so spaced as to insure thorough diffusion) through the tube, C, and the large H2O flask, where it is saturated with moisture, to the gas meter, D, where it is measured. The meter is driven by a water wheel, W, rotated at a uniform rate of speed by a constant head of water pressure, V. At E a side tube leads off from C conveying a sample of cage air for analysis. This air is drawn through a mercury valve, F, by means of the mercury pump, G. The latter is operated by the mechanism, X, Y, connected with the water wheel. From the valve F, this air passes through two H2SO4 flasks and is then saturated with moisture so as to prevent loss of water from the Ba(OH)2 tubes. Of course, the air, as it leaves the Ba(OH)2 tubes, is saturated with moisture, and is measured by the small gas meter, H, just as in the case of the large meter, while in this saturated condition. A duplicate sample is led off at the same time through another branch of the tube, E, and through another system of vessels to the gas meter at 3. Through the branches of the tube, L, duplicate samples of the air which enters the cage arc drawn in the same manner to similar valves and then through similar vessels and tubes to meters placed at 1 and 4. (Tigerstedt's "Human Physiology.")
The problem to be solved by Pettenkofer included the maintenance of a man in normal surroundings. A small room was therefore constructed which was well ventilated by a current of air. This air entered the chamber freely through an opening in connection with a large room outside and was aspirated from a second opening in the chamber, through a large gas-meter, where its volume was measured (500,000 liters per day). It was evidently impracticable to determine all the carbon dioxid in this large volume of air, but its amount was calculated from the analysis of duplicate samples continually withdrawn from the air leaving the chamber during the time of the experiment. Each sample, as it was pumped out, was made to pass over calcined pumice stone soaked in sulphuric acid, to remove the water. Next it bubbled through baryta water to remove the carbon dioxid, and then passed through a small gas-meter, where the volume of the sample was measured. After this fashion the amount of carbon dioxid and water coming from the air of the chamber was determined in duplicate. Other duplicate analyses of the air taken outside the ventilator just before it entered the chamber were simultaneously made in the same manner as were the analyses of the chamber air itself. Knowing the quantity of carbon dioxid and water entering and leaving the room, it was easy to calculate how much was derived from the man living in it during the period of experimentation. The experimenters failed to find any other gaseous exhalation from a man, such as ammonia, hydrogen, or methane, which could vitiate their results. Control experiments were made by burning a candle or evaporating a known weight of water within the room. Analysis showed that the carbon dioxid and water so produced were measurable within 1 per cent, of error.
Continue to:


