Gas Piping. Part 3
Description
This section is from the book "A Working Manual Of American Plumbing Practice", by William Beall Gray, Charles B. Ball. Also available from Amazon: Plumbing.
Gas Piping. Part 3
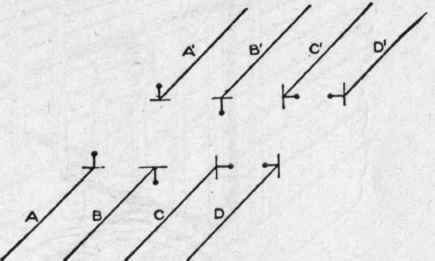
Fig. 103. Skeleton Diagrams of Bracket Pipes..

Fig. 104. Lines of Gaspipe in Frame Building, Showing How Pipes are Secured in Place.
Coal gas, and natural gas of some locations, has a strong odor that betrays leakage. Some natural gas is devoid of odor, in which case leakage is very dangerous, as there is no way quickly to detect its presence. For natural gas work, 10 pounds' air-pressure should fail to develop the slightest leak in the pipe, although the street pressure is usually even less than eight ounces. For lighting gas, the street pressure is seldom over 18 tenths water-pressure, and a 5-pound test is ample. These tests should be made with a mercury gauge, 2 inches height of column being considered as one pound pressure. A job may be considered tight when the mercury column not only does not drop, but does not even get flat at the top in from fifteen to twenty minutes' trial.
Every gas company has rules as to the number of lights allowed to be supplied from each size pipe, and the relative lengths of pipe permitted of each size. The following table gives sizes of gas pipes for different numbers of burners and lengths of runs, as usually installed:
Table IV. Maximum Run And Number Of Burners For Gas Pipes
Size of Pipe | Greatest Length of Run, Feet | Greatest Number of Burners to be Supplied | ||
3/8 | inch | 20 | feet | 2 |
1/2 | ,, | 30 | ,, | 4 |
3/4 | ,, | 50 | ,, | 15 |
l | ,, | 70 | ,, | 25 |
1 1/4 | inches | 100 | ,, | 40 |
1 1/2 | ,, | 150 | ,, | 70 |
2 | ,, | 200 | ,, | 140 |
2 1/2 | ,, | 300 | ,, | 225 |
3 | ,, | 400 | ,, | 300 |
4 | ,, | 500 | ,, | 500 |
No restrictions are observed in selecting fixtures for coal or natural gas. Coal gas carries enough carbon with it to produce a lighting flame when burned at the ordinary flame temperature. When the jet is lighted, the hydrogen is consumed in the lower part of the flame, producing sufficient heat to render incandescent the minute particles of carbon carried by it. The hydrogen, in the process of combustion, combines with the oxygen of the air, forming an invisible vapor of water, while the carbon unites with the oxygen, forming carbonic acid, or is set free as soot.
Various causes tend to render combustion incomplete. There may be excessive pressure of gas, lack of air, or defective burners. An excess of pressure at the burners causes a reduction of the amount of illumination; on the other hand, if the pressure is insufficient, the heat of the flame will not raise the carbon to a white heat, and the result will be a smoky flame. It therefore follows that for every burner there is a certain pressure (usually 5/10 of an inch water-pressure before mentioned) and a certain corresponding flow of gas, which will cause the brightest illumination.

Fig. 105. Single-Jet Burner..

Fig. 106. Bat's-Wing or Slit Burner..
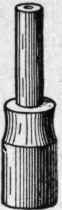
Fig. 107. Union-Jet or Fish-Tail Burner..
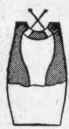
Fig. 108. Vertical Section of Union-Jet Burner..
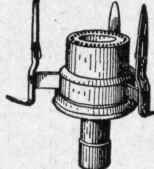
Fig. 109. Argand Burner..

Fig. 110. Lava Tip for Bat's-Wing Burner..
Burners. Many types of burners are on the market, among which the single-jet, bat's-wing, fish-tail, Argand, regenerative, and incandescent burners are the principal types.
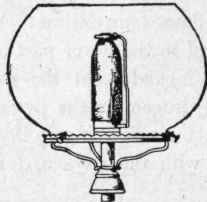
Fig. 111. Gas Burner with Globe and Incandescent Mantle.
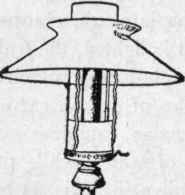
Fig. 112, Mantle Burner with Chimney and Shade.
The single-jet burner, Fig. 105, is the simplest kind, having but one small hole from which the gas issues. It is suitable only where a very small flame is required.
The bat's-wing or slit burner, Fig. 106, has a hemispherical tip with a narrow vertical slit from which the gas spreads out in a thin, flat sheet, giving a wide and rather low flame resembling in shape the wing of a bat; from which it is named.
The union-jet or fish-tail burner, Fig. 107, consists of a flat tip slightly depressed or concaved in the center, with two small holes drilled, as shown in Fig 108. Two jets of equal size issue from these holes, and, by impinging upon each other, produce, at right angles to the alignment of the holes, a flat flame longer and narrower in shape than the bat's-wing, and not unlike the tail of a fish. Neither of these burners requires a chimney, but the flames are usually encased with glass globes. They are not well suited for use with globes, however, since when one of the jets becomes choked, as it frequently does, the other is likely to crack the glass.
The Argand burner, Fig. 109, consists of a hollow ring of metal or lava, connected with the gas tube, and perforated on its upper surface with a series of fine holes, from which the gas issues, forming a round flame. This burner requires a glass or mica chimney. As an intense heat of combustion tends to increase the brilliancy of the flame, it is desirable that the burner tips shall be of a material that will cool the flame as little as possible. On this account, metal tips are inferior to those made of some non-conducting material, such as lava, adamant, enamel, etc. Metal tips are also objectionable because they corrode rapidly, and thus obstruct the passage of the gas. Fig. 110 shows a lava tip for a bat's-wing burner. Burner tips should be cleaned occasionally, but care should be taken not to enlarge the holes.
By introducing the Bunsen principle, incandescent burners give good service with coal gas. In the incandescent burner, the heat of the flame is applied in raising to incandescence some foreign material, such as a basket of magnesium or platinum wires, or a funnel-shaped asbestos wick, or a mantle treated with sulphate of zirconium and other chemical compounds. A burner of this kind is shown in Fig. 111, in which the mantle can be seen supported over the gas flame by a wire at the side. Fig. 112 shows another form of this burner, in which a chimney and shade are used in place of a globe. Burners of this kind give a very brilliant white light when used with natural or water gas. Natural gases and the so-called water gas are deficient in carbon; and, when they are used for lighting purposes, the light is produced by a burner with a mantle brought to a state of incandescence by the heat of the flame. The mantle, however, is very fragile, and is likely to lose its property of incandescence when exposed to an atmosphere containing much dust.
Continue to:


