Waterproofing Fabrics. Part 4
Description
This section is from the "Henley's Twentieth Century Formulas Recipes Processes" encyclopedia, by Norman W. Henley and others.
Waterproofing Fabrics. Part 4
The different brands of rubber vary so much, especially with regard to solubility, that it is always advisable to treat each brand by itself, and not to make a solution of two or more kinds. Oilskins and tarpaulins, etc., are mostly proofed by boiled linseed oil, with or without thickening bodies added. They are not of sufficient interest to enlarge upon in this article, so the second, or "water-repellent," class has now to be dealt with.
All the shower-proof fabrics come under this heading, as well as every cloth which is pervious to air and repulsive to water. The most time-honored recipe for proofing woollen goods is a mixture of sugar of lead and alum, and dates back hundreds of years. The system of using this is as follows: The two ingredients are dissolved separately, and the solutions mixed together. A mutual decomposition results, the base of the lead salt uniting with the sulphuric acid out of the alum to form lead sulphate, which precipitates to the bottom. The clear solution contains alumina in the form of acetate, and this supplies the proofing quality to the fabric. It is applied in a form of machine shown in Fig. 8,. which will be seen to consist of a trough containing the proofing solution, C, with a pair of squeezing rollers, A, over the top. The fabric is drawn down through the solution and up through the squeezers in the direction of the arrows. At the back of the machine the cloth automatically winds itself onto a roll, B, and then only requires drying to develop the water-resisting power. D is a weight acting on a. lever which presses the two rollers, A, together. The water-repelling property is gained as follows:
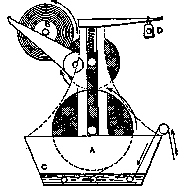
Fig. 8.
Drying the fabric, which is impregnated with acetate of alumina, drives off some of the volatile acetic acid, leaving a film of basic acetate of alumina on each wool fiber. This basic salt is very difficult to wet, and has so little attraction for moisture that in a shower of rain the drops remain in a spheroidal state, and fall off. In a strong wind, or under pressure, water eventually penetrates through fabrics proofed in this manner; but they will effectually resist a sharp shower. Unfortunately, shower-proofed goods, with wear, gradually lose this property of repelling water. The equation representing the change between alum and sugar of lead is given below. In the case of common alum there would, of course, be potassium acetate in solution besides the alumina.
Alum. Sugar of lead.
Al2K2(So4)4 + 4Pb(C2H302)2 Lead Potassium Aluminum
sulphate. acetate. acetate.
= 4PbSo4 + 2KC2H302 + Al3(C,H302)6
Now that sulphate of alumina is in common use, alum need not be used, as the potash in it serves no purpose in proofing.
There are many compositions conferring water: resisting powers upon textiles, but unfortunately they either affect the general handle of the material and make it stiff, or they stain and discolor it, which is equally bad. A large range of waterproof compositions can be got by using stearates of the metals; these, in nearly every case, are insoluble bodies, and when deposited in the interior of a fabric form a water-resisting "filling" which is very effective. As a rule these stearates are deposited on the material by means of double baths; for example, by passing the fabric through (say) a bath of aluminum acetate, and then, after squeezing out the excess of liquid, passing it through a bath of soap. The aluminum salt on the fabric decomposes the soap, resulting in a deposit of insoluble stearate of alumina. This system of proofing in two baths is cleaner and more economical than adding all the ingredients together, as the stearate formed is just where it is required "on the fibers," and not at the bottom of the bath.
One of the most important patents now worked for waterproofing purposes is on the lines of the old alumina process. In this case the factor used is rosin, dissolved in a very large bulk of petroleum spirit. The fabrics to be proofed (usually dress materials) are passed through a bath of this solution, and carefully dried to drive off the solvent. Following this, the goods are treated by pressing with hot polished metal rollers. This last process melts the small quantity of rosin, which is deposited on the cloth, and leaves each single fiber with an exceedingly thin film of rosin on it. It will be understood that only a very attenuated solution of rosin is permissible, so that the fibers of the threads and not the threads themselves are coated with it. If the solution contains too much rosin the fabric is stiffened, and the threads cemented together; whereas if used at the correct strength (or, rather, weakness) neither fabric nor dye suffers, and there is no evidence of stickiness of any description.

Fig. 9.
Fig. 9 shows a machine used for spreading a coat of either proofing or any other fluid on one side of the fabric.
This is done by means of a roller, A, running in the proofing solution, the, material to be coated traveling slowly over the top and just in contact with the roller, A, which transfers the proofing to it. Should the solution used be of a thick nature, then a smooth metal roller will transfer sufficient to the fabric. If the reverse is the case, and the liquid used is very thin, then the roller is covered with felt, which very materially adds to its carrying power. As shown in Pig. 9, after leaving the two squeezing rollers, BB, the fabric passes slowly round a large steam-heated cylinder, C, with the coated side uppermost. This dries the proofing and fastens it, and the cloth is taken off at D.
Continue to:


