Carrying Out A Fractional Distillation. Part 3
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Carrying Out A Fractional Distillation. Part 3
Third Fractionation
The third fractionation was carried out in the same manner as the second, the first portion of the first distillate, and also the first portion of the distillate which came over after the contents of receiver No. 3 had been added to the residue in the still, being collected in No. 1.
Fourth Fractionation
In the fourth fractionation, the first fraction was redistilled, the second being placed in the still when the temperature reached 81.2°. In other respects the procedure was the same as in the third fractionation.
Procedure Dependent On Number Of Fractions
If the range of temperature for each fraction had been larger, say 5° instead of 3° for the majority of them, then on recommencing the distillation after addition of any fraction to the residue in the still, the temperature would have risen at once, or very rapidly, to the initial temperature of the fraction below ; thus, on adding any fraction, say No. 4, no distillate would have been collected in No. 2, and receiver No. 3 might have been left in position while fraction No. 4 was being placed in the still and the distillation was recommenced. This method is usually adopted, but by taking a large number of receivers there is a greater amount of separation of the components in a complete distillation and, on the whole, time is saved.
Loss Of Material
On the first distillation there was a loss of 0.75 gram of material (200.0-199.25), due entirely to evaporation. In the second complete distillation the total weight fell from 199.25 to 198.2, and in the subsequent distillations the loss was in nearly every case more than 1 gram. The greater loss was due partly to increased evaporation, and partly to the small amount of liquid left adhering each time to the funnel through which the fraction was poured into the still. The total loss after fourteen fractionations was 18.1 grams, a very appreciable amount.
Relative Rate Of Separation Of Components
It will be observed that, in the first distillation, 10.9 grams of pure toluene were obtained, but that only 9.75 grams of distillate came over below 89.2°, a temperature 9° above the boiling point of benzene ; and at the end of the fourth complete distillation the weight of pure toluene recovered was 42.1 grams, while only 31.55 grams of benzene boiling within 1 degree had been obtained. It is thus evident that the separa tion of the less volatile component is much easier than that of the more volatile, and this is, indeed, always found to be the case.
It will also be noticed that the weights of the middle fractions steadily diminished, while those of the lowest and highest fractions increased ; this also invariably occurs when a mixture of two liquids is repeatedly distilled.1
1 An account has been given by Kreis * of the fractional distillation of a mixture of 25 grams of benzene and 25 grams of toluene with an ordinary distillation bulb ; the data for the first distillation are such as might be expected, but the results given for the second are quite impossible, for while in the first distillation 17 c.c. of liquid are stated to have been collected above 1.08°, in the second complete distillation the volume of the corresponding fraction is given as only 3 c.c, and it was not until the 8th fractionation that the volume again Kreis, " Comparative Investigations on the Methods of Fractional Distillation," Liebigs Annahn, 1884, 224, 259.
Weight Of Distillate Below Middle Temperature
The percentage weight of distillate, coming over below the middle temperature between the boiling points of the pure components, was not far below that of the benzene in the original mixture ; the percentage was actually 454, 47.0, 48.3 and 48.8 respectively in the four distillations, while the original mixture contained 50 per cent of benzene. Similar results are always obtained when a mixture of two liquids, which separates normally and easily into the original components, is distilled ; and, as will be seen later, when an improved still-head is used, the weight of distillate below the middle temperature should, in general, be very nearly equal to that of the more volatile component in the original mixture even in the first distillation.
Alteration Of Temperature Ranges
The results of the first four complete distillations are given in Table 38, and it will be seen that if the fractionation were continued in the same manner as before, the middle fractions would soon become too small to distil, and the first fraction would never consist of pure benzene.
It is therefore preferable gradually to increase the temperature ranges of the middle fractions, and to diminish those of the fractions near the boiling points of the pure substances.1 This was accordingly done in the subsequent fractionations, which were carried to their extreme limit. The results, together with those of the fourth fractionation, are given in Table 39.
Ratio of Weight of Fraction to Temperature Range.
So long as the fractions, or most of them, are collected between reached 17 c.c. Assuming the correctness of the data for the first distillation, the last fraction in the second should have been at least 6 times as great as that stated; it is difficult to understand how the mistake can have arisen.
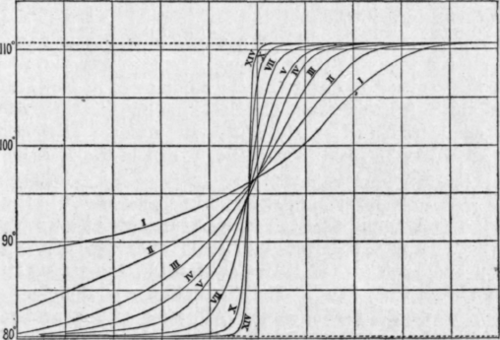
Percentage Weight of distillate.
Fig. 34. - Results of fractional distillation of mixture of benzene and toluene.
1 Mendeleeff, A paper, without title, on the fractional distillation of Baku Petroleum, Journ. Russ. Phys. Chem. Soc., Protok., 1883, 189.
Equal intervals of temperature, the weights of distillate indicate clearly enough the progress of the separation, but when the temperature ranges are gradually altered this becomes less evident, and it is advisable either to plot the percentage (or total) weight of distillate against the temperature (Fig. 34), or to divide the weight of each fraction, Aw, by its temperature range, At, and to tabulate these ratios as well as the actual weights. The purer the liquid in any fraction, the higher is the ratio Aw/At, that for a pure liquid being, of course, infinitely great; and it will be seen from Table 39 that in the later fractionations, while the weights of the middle fractions diminish, and those of the lowest and highest increase much more slowly than before, the ratios Aw/At continue to change rapidly.
Continue to:


