Chapter XXXVIII. Fractional Separation Of The Naphthas And Light Oils
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Chapter XXXVIII. Fractional Separation Of The Naphthas And Light Oils
Naphthas And Light Oils
As has been explained previously, the first distillates from the crude tar are the naphthas and light oils which come over from 80° C. to 200° C. They consist chiefly of the hydrocarbons of the benzene series ranging from benzene itself to the mes-itylenes. The separation of these is carried out entirely by fractional distillation, and, owing to the similarity in chemical composition and, in some cases, the slight difference in boiling point, the problem is by no means a simple one. Very efficient apparatus is required, and probably there is no instance where, technically, more attention has to be paid to the type of plant installed.
Products Required
The products that the distiller has to prepare in a pure state are - benzene (b.p. 80.4° C), toluene (b.p. 110.6° C), and sometimes ortho-xylene (b.p. 141.5° C), meta-xylene (b.p. 139° C), and para-xylene (b.p. 138.5° C). Generally speaking, however, the last three (if they are required at all) are made and sold as a mixture of the three isomers with a boiling-point range of 2 to 5°. Commercially pure benzene, which is used for making nitro-benzene, synthetic phenol, and other intermediates, must boil within a range of 0.5° C. and contain only small quantities of thiophene and other impurities. Pure toluene, for the manufacture of trinitro-toluene and dye intermediates, must also pass a similar specification.
Prior to the Great War neither of these products were prepared to any great extent in this country, and it was only through the urgent need of them for the manufacture of high explosives that distillers realised the importance of efficient apparatus. The quantities of benzene and toluene produced during the war were far in excess of the demands of the dye trade in normal peace times, so that there is now no further need to separate them to any large extent into the pure hydrocarbons, but they can be sold as a mixture for motor fuel. The xylenes are occasionally refined into a mixture boiling within a few degrees, where there is a demand for the production of intermediates, but generally they are sold in the form of solvent naphtha distilling 90 per cent at 160° C, a product which is a mixture of toluene, xylenes, mesitylenes, and other homologues.1
1 "By distilling 90 per cent at 160° C." is meant that, on distillation, 90 per cent of the naphtha comes over between 120° C. and 160° C.
First Distillation
The crude naphtha, or light oils whether separate or mixed, is first of all distilled in an ordinary fire still. This is similar in construction to the pot tar still already described, except that it is not generally so large and is fitted with a good fractionating column. It is found that a still of 2000 to 3000 gallon capacity is large enough for most purposes, as one of this size can usually be worked off in a day. Here again, economy can be effected by interposing a heat interchanger as condenser, in which the coil carries the vapour from the column., and the cooling medium is the naphtha for the next charge in the still. The column is of the type of any of those described later, except that, owing to the higher working temperatures., no dephleg-mator is used. The object of this first fire distillation is to make a primary split of the distillate into fractions suitable for subsequent distillations, some of which can be carried out in a steam still and some in a fire still.
Fractions Obtained
The fractions generally obtained are three, namely, crude benzole, crude solvent, and crude heavy naphtha. These, when redistilled, should have the following approximate boiling-point ranges : in the case of crude benzole 60 per cent should come over below 120° C.; in the case of crude solvent 60 per cent from 120° to 160° C. ; and in the case of crude heavy naphtha 60 per cent from 160° to 200° C. A few experiments with the plant in use will readily show the temperatures at which the changes from one fraction to another are to be made. The residues from this primary distillation are composed of creosote and naphthalene and are consequently treated in the manner already described (p. 388).
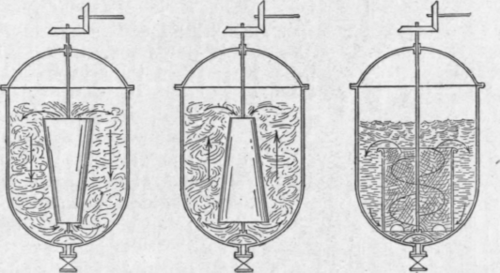
Fig. 162. - Washers.
Washing The Naphthas
Before the various fractions can be further distilled it is necessary to eliminate from them the numerous impurities they contain. The purification is carried out by violently agitating the spirit with sulphuric acid and afterwards caustic soda.
Washers
Various forms of washers or mixers are used, some of which are shown in Fig. 162. They are made either of wrought iron lined with lead, or of cast iron, and are of greater capacity than that of the stills in use, so that sufficient room is left in the washer to agitate a full still charge. Of the two metals, cast iron, in the experience of the author, has been found to be the more economical, as in practice very little action takes place between this metal and the acid. Many kinds of agitators have been devised, from the simplest air-blowing form to those illustrated, which are probably the best. A very efficient type is an elaboration of the cone agitator where two cones, one at the top and one at the bottom, are fitted on the same spindle. The washer should be so designed that the bottom is well dished or conical, in order that the last traces of acid and alkali can be drawn out without loss of naphtha.
Some distillers prefer that two washers should be employed, one in which to conduct the acid wash, and the other the caustic soda and the final washes. If the acid and soda washes are conducted in one washer, there is a great risk of some of the resinous matter, produced by the action of the acid, adhering to the sides of the washer and suffering decomposition by the caustic soda, the products then being dissolved by the spirit.
Distillates obtained from naphthas, rendered impure in this way, possess a peculiarly unpleasant odour and rapidly go off colour.
Continue to:


