Distillation Of Alcohol On The Manufacturing Scale
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Distillation Of Alcohol On The Manufacturing Scale
The Hon. F. R. Henley, M.A., F.I.C. And Oseph Reilly, M.A., D.Sc, F.R.C.Sc.L, F.I.C.
Chemist-In-Charge At The Royal Naval Cordite Factory, Holton Heath, Dorset
Chapter XXVI. Distillation Of Mixtures Of Ethyl Alcohol And Water
(theoretical)
A very large proportion of the ethyl alcohol now produced is prepared by the fermentation of sugar derived from grapes, beet, molasses, grain, or wood. The design of the apparatus used for distilling depends on the nature of the fermented liquids and the quality of the finished products required. From the distiller's point of view the substances contained in the liquids to be distilled may be divided into three classes - (1) the volatile products of the fermentation, from 1 to 12 per cent of ethyl alcohol; small quantities of the higher alcohols, amounting to 0.05 to 0.7 per cent of the raw spirit, principally iso-amyl alcohol and d-amyl alcohol, and smaller amounts of propyl alcohol; aldehydes, acids, esters, and furfurol; (2) large quantities of water; (3) the solid residue derived from the raw materials used and from the yeast. The production of alcohol, freed as far as possible by distillation from water and the by-products of fermentation, is carried out in two distinct stages: - ,
(1) Distillation proper, i.e. the extraction of the volatile from the non-volatile constituents of the fermented wash by boiling in a suitable vessel.
(2) Rectification, - The separation in as pure a state as possible, by means of fractionation, of ethyl alcohol and the other volatile products of fermentation.
Before describing the practical methods adopted for obtaining strong alcohol on the commercial scale it will be well to review the available knowledge of the principles underlying the distillation of 1. Mixtures of ethyl alcohol and water.
2. Mixtures of water and ethyl alcohol with the other volatile products of fermentation. On this knowledge is based the design and control of the stills.
Distillation Of Mixtures Of Ethyl Alcohol And Water
In general, when a mixture of ethyl alcohol and water is boiled, the vapour produced contains a higher proportion of alcohol than the original mixture. There is no simple rule connecting the alcoholic content of liquid and distillate in every case, but a great deal of research has been done to establish these relations for a large number of different mixtures and to determine their boiling points.
The results obtained by Sorel (Distillation et rectification industrielle, 1899) and Lord Rayleigh1 are shown on Table 102 and Fig. 101.
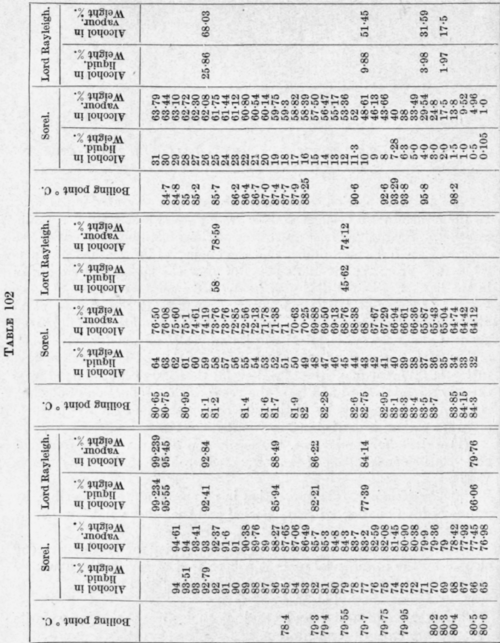
It will be observed that the ratio of alcohol in the vapour to alcohol in the liquid is very high when mixtures of low alcohol content are boiled, as is shown by the steepness of the curve on the left of the figure. But as the alcohol content of the liquid rises the curve becomes nearly horizontal, showing that there is not much more alcohol in the vapour than in the liquid boiled.
1 Rayleigh, Phil. Mag., 1902 [6], 4, S, 521.
At a point where the aqueous alcoholic liquid contains a particular weight of alcohol, the vapour evolved has the same composition as the liquid. According to Young and Fortey 1 this proportion of alcohol is 95.57 per cent by weight. Wade and Merriman 2 give approximately the same figure, i.e. 95.59 per cent. A mixture of this composition boils at a slightly lower temperature than any other mixture (see Table 103). According to Noyes and Warfel 3 there is a lowering of the boiling point of pure ethyl alcohol by 0.126°, while according to Merriman 4 the lowering is 0.15°.

Fig. 101.
By distillation alone, alcohol stronger than 95.57 per cent by weight cannot be prepared. Table 102 cannot be used directly for the calculation of the number of plates required in a rectifying column to produce spirit of any required concentration from a given mixture. For instance, if the liquid to be distilled contains 10 per cent of alcohol, the vapour evolved by this contains 48.61 per cent (Sorel) of alcohol. This, if completely condensed, will evolve a vapour containing 69 to 70 per cent of alcohol and so on.
1 Trans. Chem. Soc, 1902, 81, 717. 2 Ibid., 1911, 99, 997.
3 Amer. Chem. Soc, 1901, 23, 463. 4 Trans. Chem. Soc., 1913, 103, 628.
Table 103. Noves and Warfel's Table
Alcoholic strength. Per cent by weight. | Boiling point. | Alcoholic strength. Per cent by weight. | Boiling point. |
100 | 78.300 | 85 | 78.645 |
99 | 78.243 | 80 | 79.050 |
98 | 78.205 | 75 | 79.505 |
97 | 78.181 | 65 | 80.438 |
96 | 78.174 | 55 | 81.77 |
95 | 78.177 | 48 | 82.43 |
94 | 78.195 | 35 | 83.87 |
93 | 78.227 | 26 | 85.41 |
92 | 78.259 | 20 | 87.32 |
91 | 78.270 | 10 | 91.80 |
90 | 78.323 | 0 | 100.00 |
In practice it is not possible on economic grounds to condense the whole of the vapour supplied to each plate and then re-evaporate this. What actually happens is that only a portion of the vapour is condensed. The liquid so produced will evolve a vapour less rich in alcohol than in the former case. Concentration of the product as it rises from plate to plate will proceed more slowly than in the ideal case. As a larger and larger proportion of the original vapour evolved is condensed the nearer will the conditions approach the ideal.
When any mixture of ethyl alcohol and water is boiled, unless the percentage of alcohol is equal to or greater than that in the mixture of constant boiling point - which would never occur in practice, starting from weak spirit - the alcohol content of the liquid progressively falls and with it the alcohol content of the vapour evolved at any moment.
Continue to:


