Fractional Separation Of The Naphthas And Light Oils. Part 9
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Fractional Separation Of The Naphthas And Light Oils. Part 9
General Practice
The practice followed in distilling for pure benzene and toluene will necessarily vary with different distillers and with different types of plants. Generally speaking, however, the crude benzole is run and preliminary fractions separated. Although, with the present forms of column, it is possible to separate pure products direct from the crude material, the time occupied in eliminating intermediates is so large, and the washing difficulties are so important, that greater economy is obtained by running stills in pairs, one working crude and the other working intermediates for pure products.
Example Of Actual Results
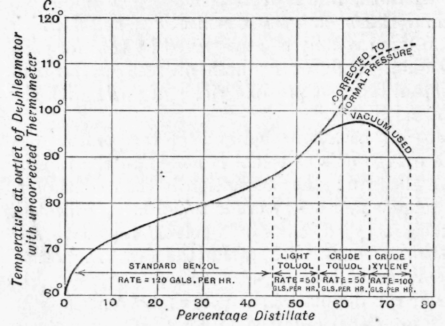
The following are some results obtained by the author using a Hodgson Thomas plant (Fig. 169, p. 406). The preliminary fractions from the crude are :-(a) standard benzole, (b) light toluol, (c) crude toluene, (d) crude xylene. The distillation is represented in Fig. 176, which shows the main features. The variation in the rate of working will be noted; the rate is rapid so long as the distillate is uniform in quality, and is slow during the passage from one fraction to another. This variation in rate is more apparent in the subsequent figures. The dotted part of the curve represents the distillation converted to normal pressure, the actual distillation under reduced pressure being shown by the solid line.
Fraction (a) consists of benzene and toluene and contains 90 to 95 per cent of benzene. It is fractionated for pure benzene and a yield of 65 per cent is obtained on the first run. The results are indicated in Fig. 177, the noticeable feature being the slow rate of distillation until the temperature at the outlet of the dephlegmator reaches a constant value ; at this stage the rate can be increased and maintained until the temperature rises, when it is advisable to stop the distillation and recharge on the residue. The temperature of the dephlegma-tor water during this period is about 42° C, which ensures a good return to the column.
Fractionation of crude benzole. Preliminary separation.
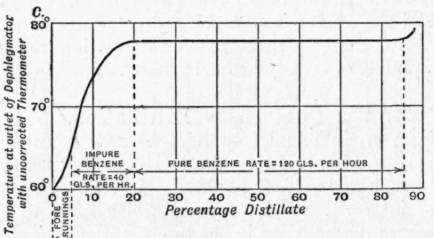
Fig. 177. - Fractionation of standard benzole.
Fraction (b) consists of about 50 per cent of benzene and 50 per cent of toluene, no xylene being present. The working of this is shown in Fig. 178. The treatment of this fraction indicates, perhaps more than any other, the efficiency of the modern column. Both pure benzene and pure toluene are obtained in one distillation from the mixture. As the temperature rises after the separation of the pure benzene the rate is decreased, and is increased again when the temperature becomes constant while the pure toluene fraction is collected. The temperature of the water in the dephleg-mator is maintained at about 42° C. on the benzene fraction, and allowed to rise slowly during the distillation of the intermediates until it reaches about 100° C. during the collection of the toluene.
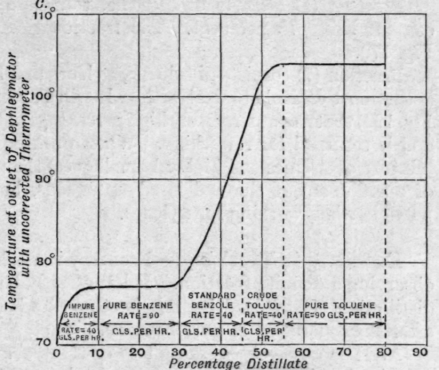
Fig. 178. - Fractionation of light toluol.

Fig. 179. - Fractionation of crude toluene.
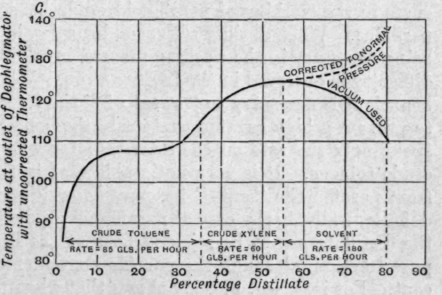
Fig. 180. - Fractionation of crude xylene.
Fraction (c) consists of benzene, toluene and xylene, the toluene content being 75 per cent. The fractionation of this is indicated in Fig. 179.
Fraction (d) consists of toluene, xylenes and higher homologues, and is the most difficult to deal with. It will be noted from the curve (see Fig. 180) that a comparatively high percentage of heavy toluol is produced and is returned for reworking. When working for solvent naphtha (a mixture of xylenes and higher homologues) the use of a vacuum or even of open steam at the end is advantageous, but should be avoided if possible when working for xylenes.
Economy In Working
Economy in working can be obtained by observing a definite rotation. It has already been pointed out that the distillation of standard benzole is stopped when the temperature rises after the separation of pure benzene.
After successively working two or three charges in this manner the combined residues have accumulated toluene and the still may then be charged with light toluol, to be followed by crude toluol. This rotation avoids unnecessary working of intermediates, thus saving both time and steam.
The rates indicated by the curves will necessarily vary with the plant and must be determined for each plant, but they indicate what has been done and show a marked improvement on the practice which obtained previous to the war.
Another point of note is the difference between the separation of benzene from benzene-toluene mixtures, and of toluene from xylene-xylene mixtures. From experiments made by the author working on pure compounds in the laboratory, it would appear that no such difficulty should exist, that is to say, there are no properties inherent in the three hydrocarbons such as the formation of azeotropic mixtures which would cause this difficulty. The cause must therefore lie in the plant, probably in the dephlegmation, and the difficulty might possibly be overcome by use of salt solution or oil as a dephlegmating medium.
Paraffinoid Crude Benzole
It may be of interest to record differences in the working of paraffinoid crude benzoles and those free from paraffins. When dealing with such benzoles, in addition to greater care being necessary in the washing process, involving greater loss, a further difficulty is observed in the fractionation. When working for standard benzole it is necessary to "sweat off " the forerunnings slowly in order to prevent a high paraffin content in the benzene, and to reduce correspondingly the rate on the intermediates when working toluene from crude toluene. It is very noticeable that in the case of products free from paraffin the "cuts" for pure benzene and toluene are clear and definite, whilst in the case of paraffinoid products a larger percentage of the distillate has to be rejected both at the beginning and end of the pure fraction, although the paraffin content may be as low as 0.5 per cent. For instance, Fig. 177 shows that when the temperature reaches 78° C. pure benzene can be at once separated and the separation con tinued until the temperature rises. In the case of a paraffinoid product, on the other hand, as much as 10 per cent at the beginning and 5 per cent at the end has to be rejected. The author attributes this difficulty to the formation of azeotropic mixtures of the aromatic hydrocarbons and the paraffins; some such mixtures are known, that of benzene and normal hexane being typical (pp. 47, 51, 215).
Another feature in the working of the paraffinoid benzole is the formation of a deposit on the coil in the still, which reduces the heating capacity to a detrimental degree. More thorough washing of the benzole does not prevent this. The nature of the deposit cannot easily be ascertained, because it is rapidly decomposed to a highly carbonaceous compound of indefinite composition. Frequent cleaning of the still is necessary to maintain the conditions required for successful working.
Continue to:


