5. Condensations. Diphenylamine From Aniline And Aniline Salt
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
5. Condensations. Diphenylamine From Aniline And Aniline Salt
Reaction:
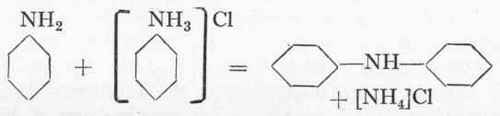
93 Gms. aniline and 93 gms. aniline hydrochloride (aniline salt) are heated for 20 hours to 230° in an enamelled autoclave fitted with an enamelled thermometer tube. The pressure reaches about 6 atms. If no enamelled thermometer tube is obtainable, it suffices simply to heat up to the requisite pressure and to note the external temperature of the oil bath, which is about 250 higher than the actual internal temperature. After 2 hours, the water present is cautiously blown off through the valve, as even traces have a very unfavourable influence on the reaction. This process is repeated three times during the course of an hour, a certain amount of aniline and ammonia also escaping. There is no point in heating for longer than 20 hours, as the only effect would be to diminish the yield. After cooling, the contents of the autoclave are placed in a porcelain dish and treated with a litre of water. The whole is then heated up to 8o° and 70 c.cs. of 30 % hydrochloric acid are added until just acid to Congo; it is then allowed to cool down over-night. The crude diphenylamine separates out as a solid cake which can be easily separated from the mother-liquor, as diphenylamine does not form a salt with the dilute hydrochloric acid. After filtering off, it is again melted up with a little water, extracted with a small quantity of hydrochloric acid and washed with dilute sodium carbonate solution.
93 gms. Aniline.
93 gms. Aniline salt.
The diphenylamine so obtained is extremely impure. It must therefore be distilled with superheated steam. For this purpose, it is placed in a half-litre distilling vessel, and the apparatus put together as shown in Fig. 17. The oil-bath is heated to 2500, and the superheater is then started up with an ordinary Fletcher burner. The water must be carefully removed from the steam, the temperature of the superheated steam being about 3000. With a good distillation it is easily possible to get over half-part base for each part water. The diphenylamine is obtained as an almost colourless liquid which solidifies to pale yellow cakes. By pouring into water it is obtained completely pure in a yield of about 100 gms.; m.p. 510. About 55 gms. aniline can be recovered from the acid mother-liquors.
Notes on Works Technique and Practice. - The autoclaves employed must be enamelled inside the cover as well as inside the vessel itself. Traces of iron or copper diminish the yield of diphenylamine by 30_50 %, resinous products being formed. The extraction with hydrochloric acid is effected in wooden vats, and the distillation by means of superheated steam is shown in Fig. 19. For superheating, modern appliances are used such as the excellent "Heitzmann Superheater," etc. It is possible to get over one part of diphenylamine with one part of water at 2300.
70 c.cs. 30 % Hc1.
β-Naphthylamine from β-Naphthol,
Reaction:

Fig. 19. - Large-scale apparatus for distillation by means of superheated steam, e.g. for a-naphthylamine, diphenylamine, etc.
(scale, 1:30).
1. Inlet pipe for superheated steam. 2. Still with slowly moving stirrer. 3. Condenser with worm.
On heating naphthol with ammonium sulphite the sulphurous ester of naphthylamine is formed. The excess of ammonia then immediately converts it into naphthylamine and ammonium sulphite.
144 Gms. (1 mol.) 100 % β-naphthol and 600 gms. ammonium sulphite are heated up in an autoclave provided with a stirrer and oil-bath.1 In addition 125 gms. of 20 % ammonia are also added. The mixture is heated for 8 hours at an internal temperature of 150o, and a pressure of about 6 atms. (N.B. steel-tube manometer). The contents are then allowed to cool, and the resultant cake of β-naphthylamine is broken up in a mortar, after which the mass is thoroughly washed out with water from a suction filter. The ammonium sulphite solution may be used several times. The well-washed base is dissolved in 1 1/2 litres of water and no gms. hydrochloric acid - which must contain no sulphuric acid - and filtered warm, a certain amount of naphthol remaining behind. The filtrate is treated with a solution of 200 gms. calcined Glauber salt dissolved in 200 c.cs. of water, the naphthylamine being precipitated as naphthylamine sulphate. It is then allowed to stand all night, the precipitate being then filtered off and well washed with cold water. For many purposes the dried sulphate is used directly (cf. p. 37).
To obtain the free base the moist sulphate is stirred up with a litre of water and treated with 60 gms. calcined soda dissolved in a little water. Owing to the sparing solubility of the sulphate, the decomposition takes several hours, but may be speeded up by continuous stirring and heating to 80o. The product is then filtered off, washed and dried at 8o°.
Yield about 130 gms. dry base, or 85-95 % of theory.
Notes on Works Technique and Practice. - For reactions of this type it is absolutely essential to use autoclaves fitted with an oil-bath or steam-jacket. The naphthylamine separates out as an oily layer at the bottom of the reaction vessel so that, in spite of stirring, if no oil-bath be used, overheating is bound to occur, leading to the conversion of a considerable portion into dinaphthylamine and decomposition products. This also holds good for the preparation of a-naphthol (see p. 102).
For technical purposes, the β-naphthylamine is usually distilled in vacuo, but great care must be taken, as it easily decomposes. If the base is not isolated, the well-dried, finely powdered sulphate mixed with 1 % of soda (cf. also Primuline) is added to the sulphuric acid or oleum as the case may be.
Continue to:


