5. Condensations. Diphenylamine From Aniline And Aniline Salt. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
5. Condensations. Diphenylamine From Aniline And Aniline Salt. Continued
144 gms. β-Naphthol. 600 gms. (Nh4)2So3 (22 %). 125 gms. 20 % Nh3.
no gms. 30 % Hc1. 1 1/2 litres H2O,
200 gms. Na2So4.
1 Ammonium sulphite is obtained by saturating 250 gms. of 20 % ammonia with SOz and then mixing the ammonium bisulphite so obtained with 250 gms. ammonia.
Bucherer's method has completely displaced the older way of heating naphthol with ammonia as this gives only 70 % yields, and requires pressure of 50-60 atms.
The Bucherer reaction may also be used for other substances, and is reversible. For example, by heating H-acid or r-acid with aniline, sodium bisulphite, and water under a reflux condenser, the corresponding phenylated amino-naphthol sulphonic acids are easily obtained, e.g.
(a) Phenyl-r-acid.
Formula:
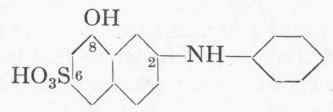
224 Gms. 100 % r-acid, 750 gms. sodium bisulphite (25 % So2), 750 c.cs. water and 200 gms. aniline are heated under a reflux for 24 hours. Sufficient concentrated sodium carbonate solution is then added to give a distinctly alkaline reaction, and the aniline is distilled off with steam. On acidifying with hydrochloric acid the pure phenyl-r-acid is precipitated. Yield about 90 %= 270 gms. 90 % acid.
224 gms. y-acid.
750 gms.
25 %
NaHso3. 750 c.cs. H2O.
200 gms. Aniline.
(b) Nevile and Winther's acid. Reaction:

Naphthol sulphonic acta 1:4. Nevile and Winther.
100 Gms. of 100 % naphthionate, dissolved in 200 c.cs. of water, are boiled for a day under a reflux with 600 gms. of sodium bisulphite solution (25 % So2). Sufficient 30 % caustic soda solution is then added to redden thiazole paper, and the whole is boiled so long as ammonia is evolved. The product is then made permanently mineral-acid with hydrochloric acid, the crystalline Nevile-Winther acid being obtained on cooling; it is separated from the residual naphthionic acid by redissolving and filtering. Yield up to 80 % of theory.
100 gms.
100 %
Naphthionic acid.
200 c.cs.
H2O.
600 gms.
25 % NaHso3 a-Naphthol from a-Naphthylamine.
Reaction:
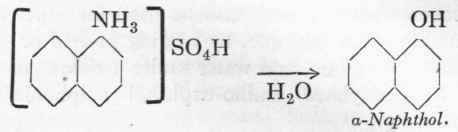
143 Gms. a-naphthylamine are mixed with no gms. 66 % Be. sulphuric acid and 1 litre of water, and the whole heated to 2000 at 14 atms. pressure. The naphthylamine should first be melted in the hot water and the acid then added in a thin stream with good stirring. The autoclave should be either lead-lined or enamelled and provided with a good stirrer; the cover may be made of iron, as the sulphuric acid is not volatile. Here also it is necessary that the autoclave should be oil-heated in order to prevent any overheating, otherwise, especially in the works, the lead will certainly be melted.
After 8 hours it is cooled down and the naphthol separated from the mother-liquor, the ammonium sulphate being recovered from the latter. The a-naphthol is melted with a little water, and after solidifying, separated from the liquid; it is almost chemically pure. To obtain it absolutely pure, vacuum distillation is resorted to. Yield, 94-95 % of theory. M.p. 94o.
Notes on Works Technique and Practice. - The process described above is the cheapest and best. There is, however, another which is analogous to the preparation of β-naphthol. The sodium salt of a-naphthalene sulphonic acid is melted with caustic soda at 290-3000. The sulphonation is carried out at 80-90o, and the salting out effected in as concentrated a solution as possible. Here also the excess of acid may be removed with advantage by milk of lime or chalk, after which the product is treated with soda, and the evaporated sodium salt melted up without further treatment. The a-naphthol so obtained is impure.
143 gms. a-Naphthylamine.
110 gms. H2So4, 66° Be.
1 litre H2O.
Dimethylaniline. (Diethyl- And Ethylbenzyl-Aniline.)
Reaction:

Plate IX.
Diphlegmating Columns

Fig. 25.

Fig. 25A.

Fig. 26.
Kubierschky Columns
Raschig Column.
Diameter of columns, 50-150 cms. Height, 8-16 cms. The upper part (1-2 metres) is externally cooled during rectification. The remainder of the column (7-15 metres) is well insulated, and the top opening is closed.
(b)
For the preparation of dimethylaniline an iron autoclave is used with a cast-iron lining, working up to 60 atms. pressure and provided with oil bath, manometer, etc. The methyl alcohol (wood spirit) used for the alkylation must contain no traces of acetone or ethyl alcohol, as the presence of such impurities leads to an immense increase in the pressure; its purity must therefore be tested by means of the iodoform reaction.
93 Gms. of pure aniline are mixed with 105 gms. pure methyl alcohol and 9.4 gms. of 94 % (66° Be.) sulphuric acid. The autoclave is then closed and the oil-bath heated to 200o; the pressure rises to about 30 atms. and the contents are then left for 6 hours at 215o. They are allowed to cool and are then treated with 25 gms. 30 % caustic soda lye. In order to split up the sulpho-ammonium bases formed at the same time (which are only decomposed at higher temperatures into sulphuric acid, alcohol and tertiary amine), the product must be heated up to 170° in the autoclave for a further 5 hours.1 The contents of the autoclave are distilled over with steam, the dimethylaniline completely salted out from the aqueous solution with common salt, after which it is removed with a separating funnel and distilled through a small bulb column. It is obtained almost chemically pure as a colourless liquid which contains, however, always some monomethylaniline.2 Yield about 117 gms. B.p. 192o.
(b) Diethylaniline.
The preparation of diethylaniline in the laboratory is also quite simple, but should only be carried out in enamelled autoclaves, as hydrochloric acid is used instead of sulphuric acid, ethyl alcohol
93 gms. Aniline.
105 gms. Ch3oh.
9.4 gms H2So4, 66° Be.
25 gms.
30 % NaOH.
1 The formation of quaternary ammonium bases is especially noticeable in the preparation of ethylbenzyl-aniline and methylbenzyl-aniline.
2 The purity may be tested by mixing 4 c.cs. of the dimethylaniline with 2 c.cs. of acetic anhydride. The temperature should not rise more than 1° at most (acetic anhydride test).
being simply split up by sulphuric acid into water, carbon, and ethylene.
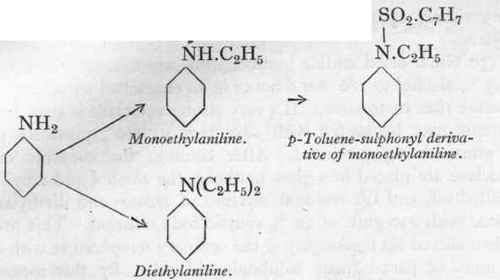
130 Gms. dried aniline hydrochloride are heated with 140 gms. of 95 % alcohol to 180o for 8 hours in an enamelled autoclave. The pressure rises to 30 atms. If a very strong autoclave is available, the contents may be heated with advantage to 2000, pressures up to 55 atms. being produced. After cooling, the contents of the autoclave are placed in a glass bolthead, the alcohol and ethyl ether distilled off, and the residual mixture of mono- and diethylaniline treated with no gms. of 30 % caustic soda solution. This product is then stirred up thoroughly at the ordinary temperature with about 40 gms. of para-toluene sulphonic chloride. By this means the monoethylaniline is converted into the toluene sulphonic derivative, which is not volatile in steam, so that the diethylaniline may be distilled over quite pure. The purity is tested by the acetic anhydride test, the sulphonic chloride treatment being repeated if necessary. Yield about 120 gms.
The residual toluene sulphonic derivative may be hydrolysed with concentrated sulphuric acid, and the monoethylaniline recovered.
Notes on Works Technique and Practice. - The heating up of a big autoclave in the works takes from 4-6 hours, and must be carried out very cautiously. As soon as the temperature has reached about 190o the pressure rises rapidly by itself to 10-30 atms. After the reaction is finished, the excess of methyl alcohol is blown off, together with the ether, the vapours being condensed. The hydrolysis of the sulpho-ammonium base is carried out in huge boilers containing from 3000-5000 kgs. dimethylaniline.
The method given above for the preparation of dimethyl- and diethyl-aniline is not very satisfactory, but may be recommended as a simple process. A cheaper and more rational method of preparation consists in using less alcohol and acid, the resultant mixture being saponified directly with caustic soda lye. The monoalkyl derivative is then converted into the alkyl benzyl derivative by means of benzyl chloride; this process is effected according to the scheme given for the preparation of Chrysophenin and nitrophenetole, or simply by heating the monoalkyl derivative in a closed vessel at 125o with the necessary quantities of benzyl chloride and 50 % caustic soda lye; 105 % of theory of benzyl chloride is needed. In this manner it is possible to arrange to obtain any required quantity of dialkyl aniline or of mixed amine. The separation is effected by means of steam distillation, the non-volatile benzyl derivative remaining no gms.
130 gms. Aniline salt.
140 gms. Alcohol.
30% NaOH.
40 gms. p-Toluene sulphonic chloride.
behind. Complete purification is effected by fractional distillation in vacuo; only absolutely pure products give the best yields of dyes of the Acid Violet or Patent Blue series.
Continue to:


