III. Technical Details. 11. Vacuum Distillations In The Laboratory And In The Works
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
III. Technical Details. 11. Vacuum Distillations In The Laboratory And In The Works
The process of distilling under reduced pressure, commonly called Vacuum Distillation, is one of the most important operations in colour technology. Certain products are distilled under reduced pressure because they decompose at their boiling point under ordinary atmospheric pressure, and also because vacuum distillation offers certain other advantages. Owing to the lower boiling point, the radiation losses are smaller and, in addition, it is often possible to heat with steam instead of with fire, so that the apparatus may be placed wherever it is most convenient without any danger of fires. In addition, there is another very important circumstance which by itself often makes it desirable to distil various substances under reduced pressure, namely, the easier separation of mixtures composed of substances whose boiling points lie close together. Thus it is only possible to effect a satisfactory separation of the three isomeric nitro-toluenes by distillation in vacuo, and the alkyl-benzyl-anilines also can only be separated easily into their components in vacuo.
Whilst the fractionating columns used in the laboratory are for the most part of older and well-known forms, on the technical scale new and complicated pieces of apparatus have gradually been evolved. The older types of columns, in which the rising vapours had to pass through a downward stream of liquid by means of which they were washed or dephlegmated, are becoming obsolete, and are rapidly being replaced by more modern and efficient columns.
The principal of these new columns is as follows: The vapours must take as long a path as possible through the downward stream of liquid, but must meet with only slight resistance. Further, the stream of liquid must be finely divided, so as to offer as large a surface as possible for the interchange of the various high-boiling substances. At the present day there are two important types: First, the Kubierschky column, in which the gases have to take a zig-zag course,
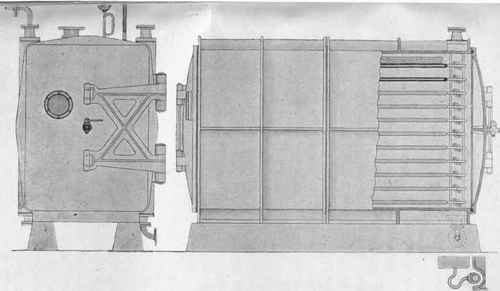
Fig. 40. - Vacuum drying chest for dyes (Passburg system) (scale 1 : 30).
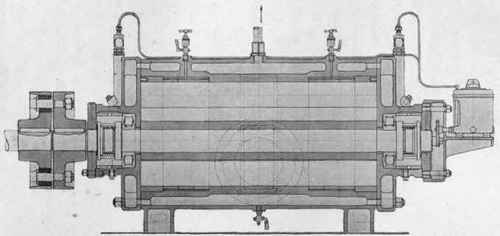
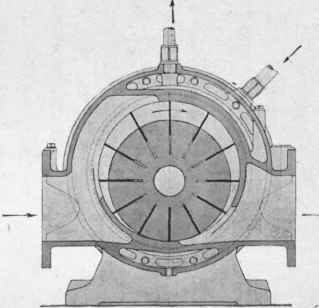
Fig. 41. - Rotary compressor and vacuum pump (made by the Swiss Locomotive and Machine Factory, Wintherthur) (scale 1 :10).
Plate XVI.
during which they meet with a descending shower of liquid. Plate IX shows, in section, two forms of this extremely efficient apparatus. They are used not only for fractional distillation, but also for the removal of ammonia from gas-liquor, for obtaining bromine from Stassfurth mother-liquors, and for many other purposes. If it is used for distillation the upper opening is closed, the top portion of the column being cooled by spraying, whilst the remainder (9/10-19/20) is insulated.
The other form of construction, due to F. Raschig, consists in a tower filled with small cylinders, of equal height and diameter. On filling a tower with such cylinders they lie irregularly, as indicated in the drawing on Plate IX, so that the surface with which the gas and liquid come in contact is very considerable.
The vacuum is produced by means of a reciprocating pump, which brings down the pressure to about 50 mms. of mercury. Lower pressures down to about 8 mms. may be obtained by running two pumps in series. But here also there has been a change during the last few years; of late increasing use is being made of modern rotating pumps, such as the excellent pumps made by the Swiss Locomotive and Machine Factory in Winterthur, diagrams of whose compression and vacuum pumps (Witte's system) are shown on Plate XVI. The method of working is indicated by the diagrammatic sketch: the movable slides enclose a definite volume of air and drive it towards the exit-opening, by which means it is compressed. The machine is reversible, and may be used either as a compressor or as a vacuum pump, pressures being obtainable with it from about 4 atmos. down to about 12 mms. The machine must be cooled, as may be seen from the drawing. It is coupled up directly with an A.C. motor, giving 1500-2500 revolutions per minute, the loss of power on the transmission being almost negligible. As already mentioned, two rotating pumps are frequently coupled up together, one behind the other.
Where it is desired to subject liquids to a vacuum distillation, an ordinary boiler is made use of, which is frequently of very large capacity. For instance, as much as 20,000 kilos, of aniline may be vacuum distilled in a boiler which is heated by means of several steam coils. The case, however, is different when dealing with β-naphthol or a similar product, and such large quantities cannot be distilled at once. Quantities of 2000 kilos, and over can indeed be dealt with, but a different type of apparatus is required. The high boiling point of /3-naphthol prevents steam being used for heating, although here also very promising experiments have been made with the
Frederking apparatus. Normally the heating must be done by direct firing, and for this purpose gas heating is the most satisfactory, owing to the ease with which it can be regulated. Very often the use of an oil-bath is omitted, but in this case there is the risk of the residual pitch becoming charred, so that it is difficult to remove it from the still, and it ceases to be of commercial value.1 On the large scale it is unnecessary to introduce air during a vacuum distillation, as "bumping" does not occur. It is, however, necessary for the whole apparatus to be well insulated, and also that all tubes in which stoppages might occur shall be easily accessible and capable of being heated; the receiver must be provided with a jacket both for heating and cooling. After the distillation is finished the residual liquid is blown out of the still through a tube into closed-in moulds to avoid the inconvenience of any escaping vapours. A large empty boiler is placed between the pump and the receiver, in order to catch the water and any sublimate which may be formed. Particularly in the case of β-naphthol, large quantities of the product are carried over as a fine snow which would stop up the pump.
Continue to:


