7. Triphenylmethane Dyes. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
7. Triphenylmethane Dyes. Continued
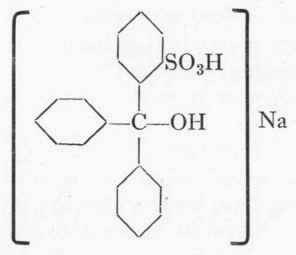
Sandmeyer was the first to recognize the connection between constitution and stability to alkalis, and his Erioglaucine, the formula of which is given below, was the first colour to be prepared in the light of this important knowledge:
Erioglaucine (Sandmeyer).
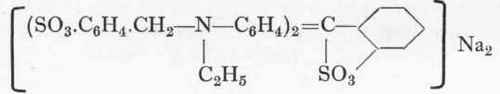
from Ethylbenzylaniline sulphonic acid and Benzaldehyde orthosulphonic acid.
Probably an internal anhydride is formed between the carbinol hydroxyl and the sulphonic group, and to this is due the great stability towards sodium carbonate and caustic soda. This hypothesis is not a mere wild guess, but is strongly supported by the fact that dyes of the formula are quite insoluble in alkalis. Reaction:

Toluene. I.
Toluene disulphonic acid 1:2:4.
II.
Benzaldehyde disulphonic acid 1:2:4.
III.
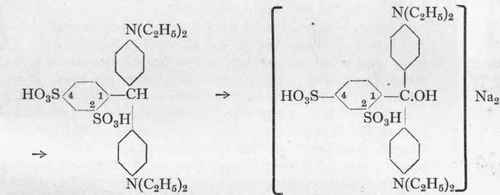
Leuco-Xylene Blue VS.
IV.
Xylene Blue VS (Steiner).
V.
(a) Toluene disulphonic acid. II.
46 Gms. pure toluene (1/2 mol.) are mixed with 80 gms. sulphuric 46 gms. acid (monohydrate) by dropping the acid into the boiling toluene 80 gms. during a quarter of an hour and then heating to 125o for an hour. 100 % The toluene will have completely disappeared by this time, and the HgSo4.
mixture is then cooled down to 30o, after which 220 gms. of 66 % oleum is run in during half an hour. The product is heated up to 125o for 4 hours, all the toluene being thereby converted into the disulphonic acid. The mixture is then diluted with 400 gms. of sulphuric acid (66° Be.) and the whole transferred to a porcelain pot provided with a good iron stirrer.
220 gms.
66%
Oleum.
400 gms.
66%
H2So4.
(b) Benzaldehyde disulphonic acid. III.
The product is treated by degrees with 125 gms. 80 % manganese paste in small portions, with good stirring.1
The addition should occupy half an hour, the temperature of the mixture being about 250. When the addition is completed, the product is stirred for a further 3 hours at 30o, and the temperature is then raised to 120o. At this temperature the mixture usually becomes so thick that stirring is impossible. The dark colour of the manganese dioxide gradually changes to a pale grey. It is rarefy possible in the laboratory to effect the oxidation so completely that all the dioxide disappears, and it is necessary to stop the reaction before this point is reached. After standing for 12 hours the mass is diluted with 2 litres water and slaked lime added until the mineral acid reaction has completely disappeared.
Litmus, however, should not be turned a pronounced blue, as any excess of alkali destroys the aldehyde sulphonic acid. The pasty mass of calcium sulphate is now treated with strong sodium carbonate solution until a filtered test-portion no longer gives a precipitate on adding sodium carbonate. The solution is filtered from the calcium sulphate and manganese, the precipitate well washed, and, if possible, the gypsum again pasted up with water and filtered. The faintly alkaline filtrate is evaporated down in vacuo to 250 c.cs., and is then filtered, if necessary, from any traces of calcium sulphate or manganese oxide. The yield may be estimated by treating an aliquot part of the solution in presence of sodium acetate with a solution of phenyl hydrazine acetate of known strength until a portion, after salting out, no longer reacts on a further addition of "hydrazine." With this reagent an intense yellow coloration is at once produced; the method of estimation is not very accurate, however.
100 gms. MnO2 as Mn3O4.
Approx. 250 gms. CaO.
About 50 gms. Na2Co3.
1 The manganese paste is calculated as MnO2,i.e. exactly 100 gms. of manganese dioxide is used in the form of the so-called "Manganese Mud," which is a waste product from the manufacture of saccharine and possesses the approximate formula Mn3O4. For estimation, see Analytical portion.
Plate I.

Fig. i. - Laboratory autoclave fitted with stirring gear. (Constructed of cast-steel. Working pressure 60 atms. Capacity, 1 litre. Weight, 33 kg. Weight of oil-bath, 11 kg.)

Fig. 33. - The stirring autoclave shown in Plate I., taken apart. 1. Liner to fix in with solder. 2. Stuffing-box. 3. Thermometer tube. 4. Stirrer. 5. Manometer. 6. Valve. 7. Body of autoclave with bolts. 8. Copper oil-bath with iron ring.
Plate XIII.
(c) Condensation to leuco compound. IV.
The whole solution is boiled up with 45 gms. sulphuric acid and 100 gms. pure diethyl aniline for two days under a reflux, after which the mixture is made alkaline by means of 100 gms. 30 % caustic soda lye, and the excess of diethylaniline is driven off with steam. If necessary the alkaline solution is filtered and is then made just acid with 50 gms. concentrated sulphuric acid. The internal anhydride of the leuco compound is precipitated in the course of 24 hours as fine white needles, which are filtered off and thoroughly washed out with water. After drying thoroughly at 8o° they weigh 70 gms.
45 gms. H2So4, 66° Be.
100 gms. Diethylaniline. 100 gms. 30% NaOH.
About 50 gms. cone. H2So4.
(d) Oxidation to the Dye. V.
The oxidation closely resembles that for Malachite Green. 50 Gms. of the leuco compound are dissolved in a boiling solution of 8 gms. anhydrous sodium carbonate, as the sparingly soluble leuco compound is practically insoluble in cold soda solution. The liquid should be perfectly neutral to litmus, and is made up to 1800 c.cs. at o°. A mixture of 15 c.cs. concentrated sulphuric acid and exactly 22 gms. 100 % lead peroxide paste is added in one operation to the mixture (cf. Malachite Green).
After standing an hour at 0-5o the product is heated to 8o° and the lead sulphate filtered off. The solution is evaporated down to 150 c.cs., preferably in vacuo, and 50 gms. salt are added. The finished dye, which comes out in a beautifully crystalline form during the course of a day, is filtered off and washed with a little saturated brine. It is then dried in a small porcelain basin, after adding a drop of ammonia to neutralize any remaining trace of mineral acid.
Yield of concentrated dye = 32 gms.
Notes on Works Technique and Practice. - The benzaldehyde disulphonic acid is so easily soluble that it is not possible to isolate it. The oxidation is effected in large kneading troughs with interlacing arms such as were first built by Werner and Pfleiderer. The apparatus may be heated by means of a steam jacket, and owing to its powerful construction the mixing may be continued right up to the end. By this means it is possible to work with rather less sulphuric acid than indicated above. Liming and evaporation are carried out by known methods, but one difficulty shows itself. The heating tubes become very quickly encrusted with calcium sulphate, and owing to the sensitiveness of the aldehyde disulphonic acid it is not possible to use any excess of soda in order to precipitate the lime
50 gms.
Leuco compound.
8 gms.
Na2Co3.
15 gms. conc.
H2So4.
22 gms.
PbO2
100 %.
completely. The condensation is carried out in homogeneously lead-lined stirring autoclaves, whilst the oxidation is done in wooden vats provided with propeller stirrers constructed of ash. The evaporation is performed in vacuo, and the separation of the well-crystallized colouring matter is done by centrifuging. On treating the mother-liquor with aniline an impure dye is formed which constitutes the Mark II. of commerce.
More recently the 1:2:4~benzaldehyde disulphonic acid is often made from p-toluene sulphonic chloride instead of from toluene. Owing to this and other uses for the substance the once almost valueless sulphochloride has risen considerably in price.
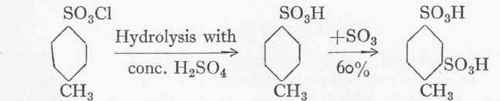
p-Toluene sulphonic chloride.
p-Toluene sulphonic acid.
1:2:4-Toluene di-sulphonic acid.
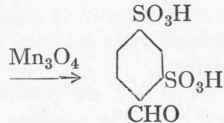
1:2:4-Benzaldehyde disulphonic acid.
Continue to:


