Chrysophenine Goo. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Chrysophenine Goo. Continued
A - N2 - B - N2 - C - N2 - D
{e.g. Naphthogene Blue 4R; the combination Naphthylamine-disulphonic acid-2:4:8 - Cleve acid-17 - Cresidine - p-Xylidine) dye cotton more or less well without the aid of mordants.
If A is an amine of the benzene series then the dye will be especially fast to light if the para position is replaced by an acetyl -amino group, or by an oxalylamino group: NH.CO.Cooh. Thus p-amino acetanilide affords products which are very fast to light. H-acid is also distinguished by the fact that azo colours obtained from it are very fast to light and particularly pure in shade (Benzo Fast Blue FF). In addition many naphthylamine disulphonic acids are of importance in this connection.
As regards B and C there are a large number of possibilities. Cleve acids 1:6 and 1:7 are used, and also m-toluidine, which differs from aniline in coupling readily with azo components, m-Amino-p-cresol methyl ether, the so-called "Cresidine," is much used, as dyes containing this component possess great purity and strength.
As to D two compounds are of special importance, namely aminonaphthol sulphonic acid 2:5:7 (J-acid) and p-xylidine. Colours derived from p-xylidine may be diazotized further on the fibre, and unite with naphthols and amines to yield products which are fast to light and washing.
As may be seen from these indications the possibilities are almost unlimited. More than a hundred dyes of this type are met with in commerce, and every colour factory places both patented and free products on the market.
(a) Aniline-Cleve acid-1:7.
9.4 Gms. pure aniline are diazotized as described on p. 108, and the diazonium solution is neutralized by means of sodium formate, which is cheaper than the acetate, until it is just mineral acid. The neutralized solution is added to 22.3 gms. Cleve acid 1:7 dissolved in 300 c.cs. water, using the pure sodium salt of the latter. As soon as the solutions have been mixed, a further 25 gms. sodium formate in concentrated solution are added, which has previously been made faintly acid to litmus by means of formic or acetic acids. The aniline combines with the Cleve acid at 8° within 5 hours, but it is advisable to allow the coupling to stand all night, as this is the only way of ensuring that a uniform dye will be produced. Next day 20 gms. caustic soda lye (30 %) is added, and the mixture allowed to stand for at least 4 hours at 200. (It is a great mistake to try to work up such couplings too quickly.) A valueless azo colour is produced which must now be diazotized further in a special manner.
9.4 gms.
diazotized Aniline and Na formate.
22.3 gms.
100 %
Cleve acid
1:7.
25 gms. Na formate.
20 gms.
30% NaOH.
(b) Aniline-Cleve acid-1:7 - Cleve acid-1:7.
The suspension of the orange-yellow monoazo dye is treated with 60 gms. common salt and 7.5 gms. sodium nitrite. Sufficient ice is then added to reduce the temperature to o°, after which 50 c.cs. concentrated hydrochloric acid are quickly added; the reaction should be distinctly mineral-acid. In the present case the diazo-tization can be carried out only in presence of sodium chloride, and it is necessary to precipitate out the sodium salt of the colouring matter in the presence of NaCl and of nitrite, as otherwise the diazo-tization is almost impossible. A similar case was discussed when dealing with the diazotization of a-naphthylamine (see p. 109).
The temperature may be allowed to rise to 12o, and the diazotization is to be regarded as complete when nitrous acid can be distinctly noticed after 2 hours.1 If necessary, a little sodium nitrite may be added. The mixture is allowed to stand all night at 10-12o, and the brown diazo compound is filtered off quickly on a large nutsch. Although it is quite stable, it must be protected from heat and light. The mother-liquor is deeply coloured and is thrown away.
The diazo compound is now stirred up with 400 gms. ice-water to a thin paste, which is then mixed with 22.3 gms. Cleve acid and 20 gms. sodium formate, exactly as described for the first coupling. The mixture is stirred for 6 hours at 5-70, and is then allowed to stand over-night, after which it is heated up to 500 and allowed to stand a further hour at this temperature. 25 Gms. of 30 % caustic soda solution are then allowed to drop in during one hour, the dye going into solution with a blue-violet colour. Unfortunately it is not possible to salt out the dye from the alkaline solution, which must be acidified. At the same time, however, various impurities are also precipitated which are carried along and accompany the finished product. After the addition of 100 gms. of salt, the product is acidified with about 50 c.cs. of 15 % hydrochloric acid, after which the dye is filtered off, pasted up with 200 c.cs. water and 25 gms. caustic soda, and completely dissolved up at 900. 7 Gms. of 100 % sodium nitrite are added to the liquid, and the clear solution is then allowed to run into a mixture of 60 gms. of 30 % hydrochloric acid, 400 gms. ice, and 300 c.cs. water, at 6o° during half an hour. Ice is added fast enough to keep the temperature constant at about 8°. The total volume at the end should be about 1 1/2 litres, and, if a pure Cleve acid-1:7 has been used, a completely clear solution of the diazo compound will result; this again cannot be precipitated, as it decomposes readily.
60 gms. NaCl.
7.5 gms.
NaNo2 (100 %).
Ice, to o°. 50 gms. 30% Hc1.
400 gms. Ice-water.
22.3 gms. 100 % Cleve acid 1:7.
20 gms. Na formate. 25 gms.
30% NaOH.
100 gms.
NaCl.
50 gms.
15% Hc1.
200 c.cs. H2O.
25 gms. NaOH.
7 gms. 100 %
NaNo2.
60 gms.
30% Hc1.
1 Such diazo compounds are deeply coloured, and it is usually not possible to test them directly for nitrous acid by means of nitrite paper. A drop, therefore, of the solution or suspension which is to be examined is placed on a little heap of salt lying on thin filter-paper. The coloured substance is precipitated out by the salt, and by pressing the reagent paper on to the reverse side of the filter-paper it can readily be seen if mineral acid and nitrous acid are present in excess.
400 gms. Ice.
300 c.cs. H2O.
(c) Aniline - Cleve acid - Cleve acid - J-acid.
The clear, deeply coloured solution of the diazo compound which has the formula:
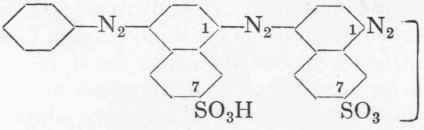
is allowed to drop during one hour into a solution of 20 gms. of 100% J-acid (aminonaphthol sulphonic acid 2:5:7), 60 gms. soda, and 300 c.cs. water. The temperature must not exceed o°, and must be regulated by means of ice. Stirring is continued for an hour, and next day the mixture is boiled up in a porcelain basin. 150 Gms. salt are then added, and the precipitate filtered off at 8o°. The mother-liquor is highly coloured and always contains a certain amount of J-acid. It is not feasible to diminish the excess of J-acid, as by so doing the yield of colour is diminished in proportion. The precipitate filters slowly, but is obtained finally almost free from salt, as it is in a good crystalline condition. It is again treated with a little 5 % brine, and is then dried at 100°, the resultant product forming a fine bronzed powder weighing about 40 gms. Benzo Fast Blue FR dyes cotton in blue shades which are fast to light and are superior to those obtained from Indigo. The fastness to chlorine, however, is very slight, and the fastness to washing only moderate. Whether the preparation has been carried out properly may be determined not only from the yield, but also from the exhaust. A correctly made dye will give exhausts of the same shade as the original dyeing, though of course correspondingly weaker.
Notes on Works Technique and Practice. - Dyes of this class are manufactured in ordinary azo-colour sheds as indicated diagram-matically on Plate VII. Owing to the instability of the diazo-compounds it is necessary to use very large filter-presses so that the whole charge may be put through in one operation. It is then possible to work up the diazo compound immediately after emptying.
20 gms. 2:5:7-acid (100 %). 60 gms. Na2Co3. 300 c.cs. H2O. 150 gms. NaCl.
the filter-press. It is also advisable to manufacture this colour during the colder season of the year, and to allow only very trustworthy men to deal with the operation. Again, it is a very good plan, whenever possible, to carry out in the laboratory the next operation, with a small portion of the intermediate product (e.g. 1/1000 th part), before the actual manufacturing stage is begun; by this means many disappointments will be avoided. The various intermediate products should also be kept as samples in a pure form so that by careful comparison one may judge whether the process is pursuing a normal course.
Continue to:


