Chrysophenine Goo
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Chrysophenine Goo
Reaction:

Brilliant Yellow.
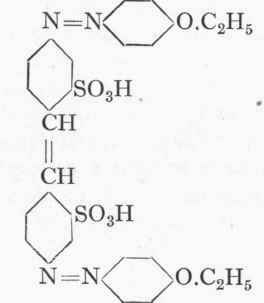
Chrysophenine Goo.
34 Gms. (1/10 mol.) of 100% diamino-stilbene-disulphonicacid is dissolved in 11 gms. sodium carbonate and 200 c.cs. water, and after cooling the acid is reprecipitated by means of 50 c.cs. (about 60 gms.) of 30 % Hc1. The temperature is reduced to 50 by means of ice, and the substance is diazotized during two hours with 14 gms. 100 % sodium nitrite. At the end a slight but detectable excess of nitrous acid should be present. Sufficient ice is now added to reduce the temperature to o° and then 20 gms. phenol, liquefied with a little water, are added. To the well-stirred suspension of phenol and tetrazo compound is added very rapidly a solution of
34 gms. Diamino-stilbene di-sulphonic acid.
11 gms. Na2Co3.
60 gms.
30%
Hc1.
14 gms. 100 % NaNo2 20 gms. Phenol.
50 gms. sodium carbonate dissolved in 200 gms. water.1 The amount of ice should be so calculated that the temperature after the addition is 8°. All goes into solution, and after a certain time a portion of the Brilliant Yellow precipitates out. After standing for 2 hours, the liquid is heated to 70°, and 100 gms. salt are added, together with enough hydrochloric acid to ensure complete precipitation of the dye, but without causing a change of colour from yellow to blue. After cooling, the product is filtered off and sucked as dry as possible at the pump. It weighs about 180 gms.
Ethylation. - The moist press-cakes are made up with water to 200 gms., and are treated with 50 gms. dehydrated sodium carbonate and 30 gms. of 35 % caustic soda lye. The pasty mixture is placed in a stirring- or rotating-autoclave, and 250 gms. 90 % alcohol are added. The autoclave is charged with 40 gms. ethyl chloride, as described on p. 74, and the mixture is then heated to 100° for 10 hours with continuous stirring (maximum pressure 6 atms.). After cooling and opening the autoclave the contents are diluted with two volumes of 10 % salt solution, and the beautifully crystalline dye is filtered off. Provided that the diamino-stilbene-disulphonic acid was free from diamino-dibenzyl-disulphonic acid, the product is about 20 % stronger than the strongest commercial colour. The yield is about 70 gms. dry concentrated colour.
Notes on Works Technique and Practice. - Chrysophenine is the most important direct yellow dye. Owing to its fastness to light upon wool, silk, and cotton, and to its low cost of production, it is almost without a competitor. In addition to the alkylation with aqueous alcohol, the lime method is also of some importance, as in the presence of lime the alkylation may be effected in aqueous instead of in alcoholic solution. In both cases it is essential that the alkyl derivative formed be precipitated at once. Which of the two processes is to be preferred depends upon the current price of alcohol. The alcohol method is better, as it gives directly a finished product of great strength which dissolves to a clear solution, and the pressure during the process does not exceed 6 atms., whereas by the lime method pressures of 25 atms. and over are encountered.
Chrysophenine gives a characteristic reaction with mineral acids, which colours it a beautiful blue. It is of scientific interest to notice that although there are no auxochromes present in the
50 gms. Na2Co3.
100 gms. NaCl. About 100 gms. Hc1.
Press-cakes 200 gms. 50 gms. Na2Co3. 30 gms. 30% NaOH.
250 gms. Alcohol. About 40 gms. Ethyl chloride. About 1 litre 10% NaCl solution.
1 Contrary to the view often held, phenol does not couple at all easily with diazo components. Diazo-ethers are frequently formed, which lead to the idea that a true azo compound has been produced. By carrying out the coupling as described for Brilliant Yellow, i.e. by first mixing the mineral acid diazonium compound with phenol (or cresol), and then adding sodium carbonate, but not caustic soda, the azo dye is obtained in far better yield.
sense of Witt's theory of colour, it is nevertheless an extraordinarily powerful dye.
The end of the alkylation of the Brilliant Yellow may be recognized in the following manner: a small test-portion is dissolved in water and treated with a few drops of acetic acid. A drop of the faintly acid solution is placed on filter-paper, and the yellow stain is touched with 10 % sodium carbonate solution. As soon as the alkylation is completed no change of colour towards reddish yellow or red should be noticeable. On the large scale samples are taken from time to time by means of a special stop-cock, and the ethyl chloride is not added all at once, but in portions of 10-15 kilos. The heating is done by means of a steam-jacket; the vessel used is a horizontal rotating autoclave with horizontal stirring gear, the stuffing-boxes of which are kept well cooled, as otherwise the alcohol dissolves out the lubricant at once. The consumption of ethyl chloride is approximately 180 % of theory.
Benzo Fast Blue FR (Bayer) from Aniline, Cleve-acid and J-acid.
Formula:

The preparation of azo colours of high molecular weight is one of the most difficult in the domain of azo chemistry. It is not possible to lay down general rules, and the following recipe is offered simply as an indication of the methods adopted. It is essential to use pure intermediate products, and the intermediate stages can only be worked up further after a preliminary purification. The dilution is often an essential point, and the sensitiveness to alkalis increases with the molecular weight, whilst the energy with which the coupling takes place rapidly diminishes with the increased size.
Almost all azo dyes of the type:
Continue to:


