Aminoazobenzene From Aniline. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Aminoazobenzene From Aniline. Continued
100 gms. Aminoazobenzene Hc1. 100 gms. Dinitrochlorbenzene. 250 gms. Na Acetate.
600 gms.
90%
Alcohol.
Fast Light Yellow G (Bayer).
Bayer's Fast Light Yellow G is the simplest member of the Pyrazolone series of dyes. These are obtained by two methods, (i) from dioxy-tartaric acid (other a-diketones are also used) and phenyl-hydrazines, (2) from phenyl-methyl-pyrazolones by coupling with diazo components. The second method is simpler, and has therefore largely displaced the older process, although large quantities of Tartrazine (from dioxytartaric acid and phenyl-hydrazine sul-phonic acid) are used at the present day. The pyrazolone is prepared from a given phenyl-hydrazine, e.g. from the phenyl-hydrazine sulphonic acid described on p. 64, and aceto-acetic ester, which is then coupled with aniline. Reaction:

i-Sulphophenyl-2-methyl-$-pyrazolone.
Fast Light Yellow G.
The hydrogen of the phenyl-methyl-pyrazolone sulphonic acid which is replaced during the coupling by the azo group is marked with an asterisk. This hydrogen atom is in the ortho position to an hydroxyl group (+) which renders possible the formation of the coupled product. It behaves exactly like the hydroxy groups in phenols and naphthols, and can bring about lake formation from azo dyes derived from ortho-amino-phenols and ortho-amino-naphthols. Dyes of the following type are produced:

In this manner Erio Chrome Red B (Geigy) is produced from i:2:4-amino-naphthol sulphonic acid (described on p. 50), and phenyl-methyl-pyrazolone (from phenyl-hydrazine and aceto-acetic ester); it is a very fast chrome wool colour:
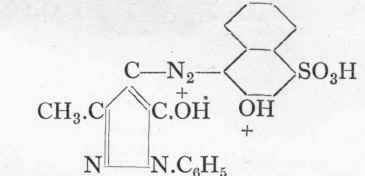
Erio Chrome Red B (Hagenbach).
(a) 1-Sulphophenyl-3-methyl-5-pyrazolone. - 19.7 Gms. 100 % phenyl-hydrazine sulphonic acid are suspended in 80 gms. of 40 % acetic acid, and to it are added 13 gms. aceto-acetic ester. The whole is then boiled up under a reflux for an hour, and is then cooled down to I5°with continuous stirring, after which the thick magma of crystals is filtered off. The yield of dry substance is 27 gms. oj 90 % pyrazolone. It is estimated by means of diazotized aniline in acetic acid solution (see Analytical Section).
(b) Fast Light Yellow G. - 326 Gms. (1/10 mol.) 100 % sulpho-phenyl-methyl-pyrazolone are dissolved in 120 c.cs. water and 6 gms. sodium carbonate, and to this are added 30 gms. sodium acetate. After cooling down to o° it is mixed with a diazo-benzene solution prepared from 9.3 gms. aniline, and the whole is stirred until a small test-portion precipitated with salt no longer gives a red coloration with alkaline resorcinol solution, which requires from 4-6 hours. The mixture is then boiled up and salted out with 100 gms. common salt. The yield is about 40 gms. strong dye.
These Pyrazolone colours, particularly the more complicated ones, are much faster than Fast Yellow (see p. 134). Dyes derived from the ortho-sulphonic acids of aromatic amines such as p-toluidine-o-sulphonic acid or p-chloraniline-o-sulphonic acid, include yellow colours which are among the fastest to light with which we are acquainted. The fastness to light can be still further increased by using chlorinated phenyl-pyrazolone for the synthesis of azo dyes, in place of sulphophenyl pyrazolones. An example of this is the Xylene Yellow of Sandoz, which is being increasingly used owing to its unexampled resistance to light, despite its relatively high price. There are, of course, almost innumerable possible modifications, of which Geigy's Polar Yellow 5G may be instanced. This has the following composition:
19.7 gms. 100 % Phenyl-hydrazine sulphonic acid.
80 gms.
40% Acetic acid.
13 gms.
Aceto-acetic ester.
26 gms. "Pyrazolone." 6 gms. Na2Co3. 120 c.cs. H2O.
30 gms.
Sodium acetate.
9.3 gms.
Aniline
(diazotized).

p-Chloraniline-osulphonic acid radical.
Aceto-acetic ester radical.
p-Amino-phenol radical.
p -Toluene sulphonic radical

Fig. 31. - Large cast-iron autoclave with steam heating (made by Gebr. Sulzer). Capacity, 1400 litres. Maximum pressure, 25 atms. Owing to the height of the stirring shaft, the water-cooled stuffing-box is easily accessible. At the top, on the right, a tap may be seen, which can be used for removing samples.

Fig. 32. - Large cast-steel autoclave (made by V. Roll). Capacity, 1500 litres. Maximum pressure, 40 atms. (The stuffing-box is too close to the bracket, which makes it difficult to pack.)
Plate XII.
Polar Yellow 5G {Richard); Swiss Cavalry Yellow.
Para-chlor-ortho-sulphophenyl-hydrazine is condensed with aceto-acetic ester, and the resultant pyrazolone coupled with diazo-p-aminophenol in acetic acid solution. The azo dye so produced, which is sensitive to alkali, is treated with p-toluene sulphonic chloride at 700 in presence of sodium carbonate and 1 molecule caustic soda lye, by which means the hydroxyl group is esterified. As a result of this ester formation the dye becomes quite fast to alkalis, and at the same time fast to milling on wool.
Notes on Works Technique and Practice. - The manufacture of the pyrazolone dyes is simple, the aryl-hydrazines being usually condensed in enamelled vessels so that as little as possible of the expensive substance shall be lost. The diazotization and coupling do not call for special remark.
Continue to:


