Aminoazobenzene From Aniline
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Aminoazobenzene From Aniline
Reaction:

Diazoaminobenzene.
Aminoazobenzene.
250 Gms. aniline are mixed with no c.cs. concentrated hydrochloric acid in a glass or porcelain beaker with good stirring, after which it is cooled down externally to 32o, and 45 gms. 100 % sodium nitrite, dissolved in a little water, are added at this temperature during half an hour. The temperature must not be allowed to exceed 340. After 2 hours the temperature is raised to 400, and after a further hour it is kept for 3 hours at 460. The mixture is now shaken out into a porcelain basin holding 250 c.cs. of water and 250 gms. ice, concentrated hydrochloric acid being then added until the reaction is distinctly acid to Congo. The excess of aniline goes into solution whilst the sparingly soluble aminoazobenzene hydrochloride remains undissolved; about 200 c.cs. hydrochloric acid are required. The hydrochloride is filtered off, thoroughly washed with 10 % brine containing 2 % hydrochloric acid, and finally with 2 % hydrochloric acid. The product is dried at 500, taking care to avoid any over-heating, as otherwise blue-black dyes, the so-called Indulines, are formed very readily by internal condensation. The yield of pure dry aminoazobenzene hydrochloride amounts to about 125 gms. For the preparation of dyes, the free base is not isolated, the hydrochloride being always used.
Fast Yellow. - Fast Yellow is the disulphonic acid of aminoazobenzene. The first sulphonic group takes up the para position to the azo group, a yellow wool dye being formed which only satisfies very modest demands as to fastness. On the introduction of a second sulphonic group, which is forced to take up the ortho position to the amino group (or azo group), the fastness to light is increased to a remarkable extent.
This sulphonation is carried out very simply: One part aminoazobenzene hydrochloride is added to three times its weight of 25 % oleum with stirring at 25° until a test portion dissolves easily in sodium carbonate. The temperature is then raised to 400, the stirring being continued, and is heated until a portion dissolves completely in a large excess of water; this will require about 5 hours. The finished sulphonation mixture is then poured on to six times its weight of ice, after which the mono-sodium salt of the disulphonic acid is salted out with 200 gms. common salt. The flesh-coloured precipitate is filtered off, thoroughly washed with 15 % brine, and the filter-cakes then stirred up with a little water. Sodium carbonate is added at 500 until the colour becomes a pure yellow, a greater or lesser quantity being required according to the thoroughness of the washing. Fast Yellow cannot be salted out, but is evaporated directly to dryness no c.cs. conc. Hc1.
250 gms. Aniline.
45 gms. 100 %
250 gms. H2O.
250 gms. Ice.
200 c.cs. Hc1 (approx.).
below 900. The yield amounts to about 200 % on the starting material.
On the large scale aminoazobenzene is prepared in large enamelled vessels holding 300-400 litres. The acidification is carried out in ordinary wooden tubs, and the mother-liquors are worked up for aniline with the aid of line and steam, the loss amounting to about
In spite of opinions to the contrary, Fast Yellow is not so fast to light as Tartrazine, and is much less fast than those pyrazolone colours which possess a sulphonic group ortho to the azo group. Aminoazobenzene is an important intermediate for many disazo dyes: if it is diazotized and allowed to act upon phenols, naphthols, and other coupling components, secondary disazo dyes are produced, the earliest of which (aminoazobenzene sulphonic acid →β-naphthol) was the so-called Biebrich Scarlet. For this reason such secondary disazo colours are termed dyes of the Biebrich Scarlet type. The diazotization of aminoazobenzene takes several hours; the freshly prepared aminoazobenzene hydrochloride is suspended in 5 parts of water and a further 150 gms. hydrochloric acid is added for each molecule of hydrochloride. It is also necessary before carrying out the actual diazotization to estimate approximately how much sodium nitrite will be required, by working up a small test portion at great dilution. The diazotization of aminoazobenzene is effected at 10-14o, and often takes a whole day on the large scale. The finished diazotized product is then either worked up at once or cooled down to o° by means of ice.
Owing to the presence of the amino group, aminoazobenzene may be condensed with dinitrochlorbenzene exactly as is the case with aniline; the product is Benzene-azo-dinitro-diphenylamine, which is a beautifully crystalline and almost insoluble substance. This can easily be converted into the monosulphonic acid by means of sulphuric acid monohydrate, a nitro-azo dye being produced which has exactly the same empirical formula as Weiler-ter-Meer's Azo Yellow, described on p. 131. It is, however, distinguished from this by its complete homogeneity, and does not split off any nitrous acid on boiling. For this reason some silk dyers prefer it to ordinary Azo Yellow, although its price is somewhat higher.
Azo Flavine FF. (B.A.S.F.)
Reaction:
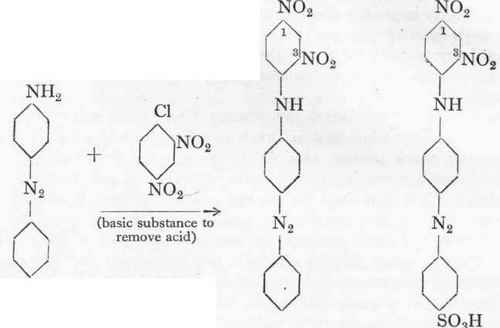
Phenyl-azo-dinitro-diphenylamine.
Azo Flavine FF.
(a) Condensation of Aminoazobenzene with Dinitrochlorbenzene. - 100 Gms. of 100 % still moist aminoazobenzene hydrochloride, 100 gms. dinitrochlorbenzene, and 250 gms. crystallized sodium acetate are heated up with 600 gms. alcohol (90 %) under a reflux for 6 hours with stirring. The condensation product separates out in the form of reddish brown, glistening crystals which are filtered off hot and washed with a little alcohol. The crystals are dried at 100°, the yield being about 115 gms.
(b) Sulphonation. - One part of the condensation product is added to three parts monohydrate and stirred for 1 hour at 300, after which the temperature is raised carefully to 45°. After 1-2 hours a test portion should give a clear solution in dilute sodium carbonate. The product is now poured into six times its weight of water, and the dye is salted out. It is filtered off acid, washed with 15 % salt solution, and then dissolved in a little hot water with the requisite quantity of sodium carbonate. The solution is salted out with 15 (volume) per cent. of salt, a gelatinous precipitate of the sodium salt being first obtained, which, however, soon becomes beautifully crystalline and easily filterable. The yield from 100 gms. condensation product is about 125 gms. strong dye. Azo Flavine FF has the shade of the lower nitrated Tropaeolines, and the great resistance to acids of the highly nitrated Azo Yellow.
Continue to:


