Mixture Of Aminoazo- Toluene And Aniline. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Mixture Of Aminoazo- Toluene And Aniline. Continued
I will give a few typical examples in order that the would-be colour-chemist may gain some idea of the importance of this aspect of the problem. In the short descriptions which have been given in this book we have become acquainted with various substances in the preparation of which always more than one main reaction product is formed. In this connection we are not concerning ourselves with inorganic by-products such as sulphurous acid, Glauber salt, or thiosulphate, but only with those organic compounds which it is customary to regard as the actual intermediates for the dye industry.
For example, in the manufacture of Cleve's acids and of the naphthylamine sulphonic acids 1:8 and 1:5 there is always a fairly constant ratio between the various isomers, which all have to be utilized. Only very occasionally is one forced to cast part of the reaction product aside until some satisfactory and profitable method of utilizing it is evolved. In the case of the acids mentioned above the position is very simple, as in the event of stocks of one of them accumulating, cheap azo dyes are produced with the aid of β-naphthol which may be either sold as such or in Black mixings.
The situation is not so simple in the case of o- and p-toluidine. When Safranine was utilized in very large quantities such accumulations of p-toluidine were obtained that the discovery of dehydrothio-p-toluidine came quite as a relief. Then, with the diminution in the demand for Safranine, such stocks of o-toluidine collected that cheap p-toluidine could only be obtained from the aniline-oil factories on condition that a certain quantity of o-toluidine was also taken, the price of which sometimes sank below 50 centimes. In a similar way the price of m-toluidine also fell below 1 franc per kilo., owing to the absence of demand. Then again, for a certain period, the toluidine question lost its importance as trinitrotoluene could be made from the o-nitrotoluene, which was used in large quantities as a shell-filling.
Another similar case is that of 0- and p-nitrochlorbenzene; for a long time the demand for p-nitrochlorbenzene was far in excess of that for the ortho compound. Then Sulphur Black T required such quantities of dinitrochlorbenzene that all the mononitro product was readily absorbed. The increasing competition in Sulphur Black T made the problem again acute, and large quantities of ortho-nitro-chlorbenzene were amassed until the production of o-nitroanisole -
- for the preparation of o-dianisidine caused so great a demand for the ortho product that it became difficult to find sufficient use for the para product. Possibly the method for the preparation of p-nitraniline given on p. 72 may offer a way out of the dilemma.

Analogous cases are the simultaneous production of R-salt and G-salt, the formation of ortho- and p-nitrophenol, and so on. In many cases it is possible by suitable alterations in the method of manufacture to suppress one product (compare for example the two methods for the preparation of amido-G-salt given on pp. 35 and
36).
Again, aminonaphthol sulphonic acid 1:8:4, for instance, may be obtained via the naphthylamine sulphonic acid 1:8 (p. 30, III), or directly from naphthalene by disulphonation and nitration.
Reaction:

A certain quantity of the β-nitro acid is formed at the same time which yields naphthylamine disulphonic acid 2:4:8 on reduction. -
Other substances also may in time accumulate in such quantities that it becomes essential at last to make use of the ever-increasing waste-dumps, as only rarely is it decided to get rid of by-products simply by burning them. An interesting case of this is the neat method worked out by C. Mettler for utilizing the o-chlorbenzoic acid which is obtained in considerable quantities as a by-product in the manufacture of o-chlorbenzaldehyde. The resinous masses of crude o-chlorbenzoic acid finally attained the dimensions of a small hill, so that it was estimated that about 30,000 kilos. of pure o-chlorbenzoic acid remained unused for years. Attempts to convert the acid into anthranilic acid by treatment with ammonia and a little copper, or a cupric salt, by Ullmann's method, showed that the well-known B.A.S.F. method (from phthalic acid via phthalamide and oxidation with the exactly calculated quantity of sodium hypochlorite by A. W. Hoffmann's method) gave a much cheaper and purer anthranilic acid.
C. Mettler's device consisted in obtaining the hitherto unknown azo-salicylic acid by a roundabout way from o-chlorbenzoic acid, as it was certain from L. Oswald's experiments that this substance would possess remarkable dyeing properties. This interesting process is effected in the following way: nitrochlorbenzoic acid is obtained by the nitration of the o-chlorbenzoic acid; this is reduced with zinc dust in neutral solution to aminochlorbenzoic acid which is diazotized and coupled with salicylic acid and the chlorine of the valueless azo dye replaced by heating to 135o with caustic soda lye and a little copper oxide. In this manner azo-salicylic acid is obtained, the chrome lake of which is distinguished by its remarkable strength and great fastness towards light, milling and potting; it also levels admirably and has therefore become a very welcome addition to the pattern-card. The following formulae will illustrate the reactions involved:
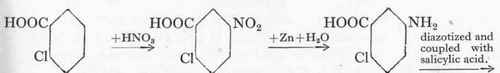
o-Chlorbenzoic acid,
Nitrochlorbenzoic acid.
Aminochlorbenzoic acid.

Valueless azo dye.
Azo-salicylic acid = Erio Chrome Flavine A.
It is not possible to obtain this compound by coupling aminosalicylic acid with salicylic acid, as this combination can only be effected directly to a very small extent (L. Oswald, v.s.).
These few, though characteristic examples, will suffice to show that with many dyes new problems will constantly occur according to the demand, which may in some cases take years to solve.
Continue to:


