Benzidine, o-Tolidine, o-Dianisidine
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Benzidine, o-Tolidine, o-Dianisidine
18.6 Gms. (1/10 mol.) of technically pure benzidine are dissolved in 23 c.cs. of 30 % hydrochloric acid and 100 c.cs. water at 700.2 The solution is cooled to 30-400 when 50 gms. ice are added, a portion of the hydrochloride being precipitated. A further 23 c.cs. hydrochloric acid, diluted with a little water, are then added with good stirring, a fresh quantity of the salt coming out. 70 C.cs. of 20 % sodium nitrite solution are then run in rapidly within 10 seconds. The temperature is about 10-120, and the solution should become clear in one minute. If the temperature has been kept too low the last traces of benzidine sulphate may remain until 8 or 10 minutes have elapsed. The solution is tested in the usual way with Congo-and nitrite-papers; it is practically neutral, but no diazo-amino . compound is formed in this case as with aniline.
1 Sulphonic acids which are already in the form of their salts are, of course, merely dissolved in water.
2 To obtain quite clear solutions, it is necessary to use hydrochloric acid free from sulphuric acid.
Tolidine and dianisidine must not be boiled, but are best dissolved below 400, or the finely divided substance is stirred up to a paste with water. In the works the solutions, together with half the hydrochloric acid, are allowed to stand over-night, the ice and remainder of the hydrochloric acid being added next day.
The Coupling of an Azo Component.
Diazo compounds are distinguished as strong or weak coupling substances according to whether they combine with salicylic acid or not. In many cases the combination may be brought about by the use of a large excess of soda or caustic soda lye, though often even this expedient fails, as many diazo compounds, indeed, are decomposed by alkali before the coupling takes place, and in these cases it is necessary to use sodium acetate or formate to combine with the mineral acid which is set free. Generally speaking it may be said that the diazo solution must be run into the phenol or amine; there are very few exceptions to this rule.
A general scheme for coupling is given here which may, in many cases, be made use of just as it stands.
1/10 gm.-molecule phenol (naphthol, amino-naphthol sulphonic acid, etc.) is dissolved in 15 c.cs. of 30 % caustic soda solution and 25 gms. sodium carbonate, together with the necessary quantity of water, and the whole cooled with ice to o°. The more concentrated the solution, the more smoothly will the coupling take place; the more acid used for diazotizing, the more alkali will be necessary.1 The diazonium solution (diazo compound) is allowed to run in a thin stream into this cold solution, the whole being stirred gently for 1 hour at a low temperature. The temperature is then raised to 300 in the course of an hour, the liquid is allowed to stand over-night, and next day the dye is separated out at a suitable temperature.
The conditions necessary for separating out the dye are very varied: it may either be filtered off in the cold directly after coupling, or it may be heated up and taken into solution, and then reprecipitated either by salting out or acidifying. In rare cases it may be impossible to separate out the dye, and one is forced to evaporate down the whole solution to dryness.
In the case of amines the scheme for the coupling needs modification in that the base is dissolved in acid (hydrochloric, acetic, or formic acid), and the alkali is replaced by acetate or formate. In very rare cases no addition is necessary, as the coupling takes place spontaneously with elimination of mineral acid. Certain amines insoluble in water, such as diphenylamine, cresidine, a-naphthylamine, etc., are sometimes coupled in alcoholic solution (cf. Tropseolin).
1 Very strongly acid diazonium solutions are neutralized with soda before coupling.
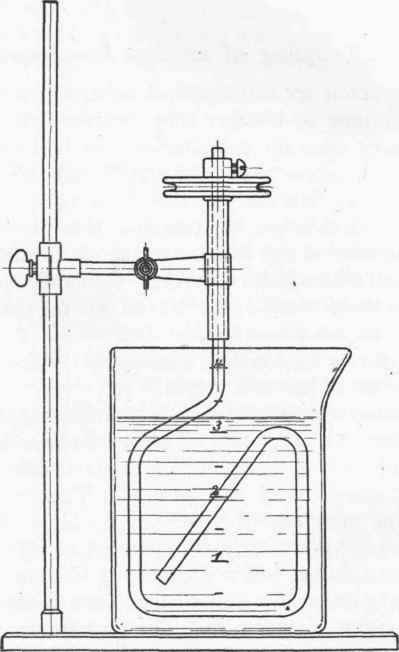
Fig. 21. - Calibrated vessel for coupling.
The pot illustrated above (Fig. 21) is specially suited for coupling. The rough graduation makes it easier to estimate the quantity of fluid present.

Fig. 28. - Arrangement of plant for vacuum distillation of substances which solidify readily (Naphthols, Phenylenediamine, etc.). 1. Distilling vessel. 2. Receiver with steam- or cooling-jacket. 3. Steam-heated tubes to prevent solidification. 4. Empty vessel for water and sublimate. 5. To pump.
Plate X.
Examples of Simple Alkaline Couplings. Acid Orange A or Orange II.
Formula:

173 Gms. (1/10 mol.) of 100 % sulphanilic acid are dissolved in 200 c.cs. water and 6 gms. soda, any excess of aniline present being driven off with steam by boiling. After filtering, 30 c.cs. concentrated hydrochloric acid are added, and the whole cooled down to 200. By means of a little ice, the temperature is brought down to 10°, and the liquid is then diazotized below 15o with 7 gms. 100 % sodium nitrite until a permanent reaction is given with nitrite-and Congo-papers.
At the same time 14.2 gms. (1/10 mol.) β-naphthol are dissolved in 15 gms. of 30 % caustic soda solution, 25 gms. soda, and 200 c.cs. water; the β-naphthol should dissolve to a clear solution. The naphthol solution is cooled to 30 with ice, and the suspension of diazo sulphanilic acid added in a thin stream, the temperature being kept below 8°. After the lapse of 1 hour, the dye formed is heated to boiling in a porcelain basin over a bare flame, and the boiling solution is treated with 100 gms. common salt added by degrees. The precipitate, which never goes completely into solution, now separates out completely, and can easily be filtered at 500 on a large suction funnel. The filter-cakes are squeezed in a screw-press, after which they are dried at 100°. The yield is about 50 gms., but can only be determined exactly by making comparative dyeing tests.
Continue to:


