Benzidine, o-Tolidine, o-Dianisidine. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Benzidine, o-Tolidine, o-Dianisidine. Continued
Notes on Works Technique and Practice. - Owing to its cheapness and brilliant shade, Acid Orange A is one of the most widely used monoazo dyes. In the works the coupling is carried out in very large pitch-pine tubs holding 15,000 and more litres, or in concrete vats lined with pieces of earthenware, holding up to 40 cubic metres. The illustration on Plate VII. shows the general works arrangement with the diazotizing and coupling tubs, together with the pressure vessel (Montejus) and the filter-press. The filtered colour is not pressed hydraulically, but is blown through in the filter-press with compressed air for 1-3 hours, and is then dried directly on copper trays. For this purpose vacuum drying ovens are coming more and more into use, as they not only facilitate rapid drying, but also afford considerable protection to the dye. The calculations for this colouring matter are given in detail later on (q.v).
17.3 gms.
6 gms. Soda. 200 c.cs. H2O.
30 c.cs. Hc1 (30 %).
7 gms. NaNo2 (100 %).
14.2 gms. β-Naphthol
15 gms.
30%
NaOH.
25 gms. Na2Co3. 200 c.cs. H2O. Ice.
Acetyl-H-Acid and Amidonaphthol Red G,
Formula:

34.1 Gms. (1/10 mol.) of 100 % H-acid are dissolved in 200 c.cs.
water and 5.8 gms. soda, at 700. To this is added within about 20 seconds, 17 gms. acetic anhydride with vigorous stirring, by which means the amino group of the H-acid is completely acetylated. The completeness of the acety-lation is determined by acidifying a small test portion of the solution with hydrochloric acid, adding a few drops sodium nitrite solution, and then mixing with an alkaline solution of H-acid. If no unchanged H-acid is present, no coloration will be produced, as no diazotizable group will be available.
This compound may be coupled with various diazo components to give beautiful azo colours which are very fast to light, and which possess excellent levelling properties; with diazotized aniline, for instance, the important Amino-naphthol Red G is formed. As the acetyl group is somewhat easily hydrolysed, it is necessary to carry out the coupling with very little soda, and, in addition, to have some ammonia present, which volatilizes when the finished colour is dried, without altering the latter.
34.1 gms.
100 % H-acid. 5.8 gms. Na2Co3.
H2Oat
70°C.
17 gms.
Ac2O.

Fig. 22. - Laboratory vacuum filter (Nutsch).
9.3 Gms. aniline (1/10 mol.) are diazotized as described, and the diazonium solution is then mixed with the ice-cold acetyl-H-acid, to which 15 gms. of 100 % soda has previously been added. After 1 minute, 20 c.cs. concentrated ammonia are added, drop by drop, and the mass is allowed to stand for 12 hours, after which the dye is salted out in the cold with 20 % of salt, calculated upon the volume of the liquid. The dye is filtered off, well pressed in the screw-press, and is dried at 500. Yield about 50 gms.
By the use of amino-acetanilide (cf. p. 71), the beautiful bluish Aminonaphthol Red 6B is obtained, which is even faster to light than the G brand.
Notes on Works Technique and Practice. - The dyes described have largely displaced the analogues from chromotropic acid (di-hydroxynaphthalene disulphonic acid 1:8:3:6), owing to their greater cheapness and fastness to light.
It is of interest to note that it is not feasible to carry out the acetylation on the works in wooden vessels; if such be used, particularly with pitch-pine, the shade of the finished product is nearly always dull. For this reason the acetylation is carried out in enamel vessels; in practice, also, a somewhat smaller proportion of acetic anhvdride is made use of.
9.3 gms. Aniline.
15 gms. Na2Co3. 20 c.cs. 20 % Nh3.
20 % NaCl.
Acid Anthracene Red G and Chromocitronine.
Formulae:
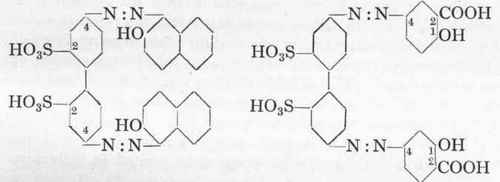
Acid Anthracene Red G (A.G.F.A.).
Chromocitronine (D .H.).
Benzidine disulphonic acid is so sparingly soluble that it must be diazotized indirectly; for this purpose it is dissolved in sodium carbonate or caustic soda solution, and the sodium salt mixed with sodium nitrite is run into the acid.
Acid Anthracene Red G.-32 Gms. benzidine disulphonic acid (100 %) are dissolved in 300 c.cs. warm water and 11 gms. soda, and the solution is then mixed with 14 gms. sodium nitrite (100 %)
32 gms. 100 %. Benzidine disulphonic acid.
at 20°. This solution is allowed to run into a mixture of 60 c cs of 3° % hydrochloric acid, 200 c.cs. water, and 100 gms. ice; the temperature may be allowed to rise to 25° without danger. The diazotization is complete in a few minutes. The tetrazo compounc is added to 30 gms. β-naphthol dissolved in exactly the same proportions of water, caustic soda, sodium carbonate and ice, as giver under Acid Orange A. The further working up is also carried out as given under Acid Orange. It may, however, happen that the tetrazo benzidine disulphonic acid separates out as a sparingly soluble, coarsely crystalline precipitate which will not couple with the alkaline naphthol solution. In this case it is necessary to treat the tetrazo compound at o° with sufficient caustic soda solution to form the soluble sodium diazotate; this couples instantaneously with the β-naphthol. Acid Anthracene Red G is fast to milling on wool without the use of mordants.
Chromocitronine. (D.H.). - The tetrazo solution of the benzidine disulphonic acid is added to a solution of 32 gms. pure salicylic acid dissolved in 80 gms. sodium carbonate and 200 c.cs. water at 5o. After 12 hours the dye which is formed is salted out in the cold with 20 % of common salt, after which it is well pressed and dried at 6o°.
In this case it is unnecessary to redissolve any of the tetrazo compound which may separate out under certain conditions as the Chromocitronine itself goes into solution at once. It is, however, very desirable, especially on a large scale, to filter the solution of the finished colour before salting out, in order that the printing rollers may not become smeared when it is used for calico printing, in which it finds extensive application. The wooden tubs always splinter off to a certain extent, which is liable to cause a good deal of trouble to the printer. Chromocitronine is a beautiful yellow dye, the chrome lakes of which are distinguished by their fastness to light, washing, and chlorine. Owing to its great solubility it penetrates well into the material so that thin fabrics appear to be printed on both sides.
300 c.cs. H2O. 11 gms. Na2Co3.
14 gms. 100 % NaNo2. 60 c.cs.
30%
Hc1.
30 gms. 0-Naphthol. 30 gms. 30% NaOH.
50 gms. Na2Co3.
32 gms. Saiicylicacid. 80 gms. Soda.
Bismarck Brown G and R.
Formulae:
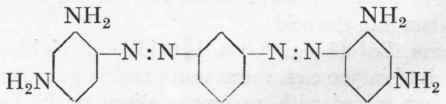
Bismarck Brown G

Bismarck Brown R.
These dyes are mixtures of various colouring matters, in which, however, the products indicated above greatly predominate. The recipes given in the literature on the subject are not very satisfactory, as they always describe the treatment of an acid diamine solution with sodium nitrite. It is, however, far better to acidify cautiously the neutral, mixed solution of the diamine and nitrite, or to allow the neutral mixture to run into the requisite quantity of hydrochloric acid during 12 minutes; the former method will be described here. Somewhat more nitrite is required than corresponds to the equation:
3 Diamine+2NaNo2 + 4 HCl = 1 Dye + (2HCl)+2NaCl.
In the case of m-phenylene diamine the excess is about 24 %, and in the case of toluylene diamine about 20 %, i.e. one uses for 3 mols. diamine, 2 mols. nitrite+20 % or 24 % excess, respectively. During the formation of the dye the diamine base disappears completely in both cases, as may be seen by salting out a test portion.
Bismarck Brown R (Vesuvine R, etc.).
36.6 Gms. pure toluylene diamine are dissolved in 1 litre of water at 400, and after cooling the solution is mixed with 16.5 gms. 100 % sodium nitrite. The volume is then made up to 1600 c.cs. with ice and a mixture of 60 gms. strong hydrochloric acid and 60 c.cs. water is then run in under the surface of the liquid with continuous stirring during 20 minutes. The solution becomes deep brown at once and evolves a fair amount of nitrogen. The end temperature is about 10°. After 8 hours, the product is salted out, with 300 gms. salt, and after standing for 3 hours the mass is filtered, the extremely soluble dye being washed on the filter with its mother-liquor. It is dried at a low temperature (in the works in vacuo), the dry product weighing about 50 gms.
The dyeings upon tannined cotton are fast to washing, cheap, and strong, but are not fast to light. In spite of this, however, both brands of Brown are much used for cotton and silk, and especially for leather. Mixed dyeings with Azo Yellow afford good brown shades upon upholstery leather, which are fast to light and rubbing.
36.6 gms.
m-Toluylene diamine.
16.5 gms. NaNo2. 1600 c.cs. H2O.
120 gms.
15% Hc1.
300 gms. NaCl.
The G mark is made in a precisely similar manner, but usually it does not come out in a good crystalline form, and is therefore troublesome to filter; this disadvantage can be removed to a certain extent by using a larger excess of nitrite.
Continue to:


