Phenylhydrazine Sulphonic Acid From Sulphanilic Acid. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Phenylhydrazine Sulphonic Acid From Sulphanilic Acid. Continued
Other nitro compounds must be reduced in ammoniacal solution with exactly the calculated quantity of hydrogen sulphide. It happens frequently that mere careful weighing of the hydrogen sulphide is insufficient, so that it is necessary to make use of various devices. Thus dinitro-phenol can only be satisfactorily reduced to nitro-amino-phenol if it is used in the form of its very finely divided sodium salt, as obtained directly by the hydrolysis of dinitro-chlor-benzene (see Sulphur Black T), which is reduced in ammoniacal solution at about 6o° with exactly the calculated quantity of hydrogen sulphide. The nitro-amino-phenol so obtained is then best re-crystallized from boiling water.
100 gms. Dinitro-benzene.
245 gms
Na2S
+9H2O
500 gms Ice.
The combination of the sparingly soluble diazo compound with the m-phenylene-diamine sulphonic acid, described on p. 46, affords the cheapest chrome-brown on the market. The coupling is effected in a completely neutral solution, and as concentrated as possible, at 280 during 2-3 days. Chrome Brown R (Kalle):

m-Phenylene-diamine from m-Dinitro-benzene.
Reaction:

For the preparation of m-phenylene-diamine the H-acid apparatus shown in Fig. 5 is made use of, and is charged with 168 gms. m-dinitro-benzene. As the commercial m-dinitro-benzene is not pure it is never possible to obtain yields higher than 90 % of theory. Commercial dinitro-benzene has a melting-point of about 80°, and always contains varying percentages of isomeric products, together, usually, with some dinitrophenol which is easily recognized by the more or less intense yellow colour which it gives on boiling with soda or with caustic soda-lye.
1 1/2 Litres of water and 300 gms. fine iron turnings are placed in the reduction vessel; the iron is etched with 20 c.cs. conc, hydrochloric acid, and the mixture heated to boiling for at least 5 minutes. The dinitro-benzene is then added in small portions of not more than 2 gms. at a time with continuous stirring. It will be noticed that the liquid first becomes yellow, due to the formation of m-nitraniline; it froths up on each addition, sometimes so vigorously that it becomes necessary to spray water on to the surface. For the reduction to go properly the temperature must always be kept up to the boiling-point. After every second addition of dinitro-benzene it is necessary to wait until a drop on filter paper is colourless. If the process be hurried too much the liquid becomes brown, due to the formation of azoxy compounds. These prevent the progress of the reduction, which must then be regarded as a failure, and must be stopped. This phenomenon is one of the most undesirable which takes place in any reduction process. It occurs also if poor quality iron be used, for which reason it is most necessary before purchase always to test the samples of iron very carefully as to their activity. With a little practice, however, it is easy to reduce the dinitro-benzene satisfactorily in 40 minutes. At the end a solution is obtained which is frequently either colourless or pale brown and darkens on keeping; it is then boiled for at least 5 minutes, the water which evaporates being replaced so as to keep the volume at about 2 litres, corresponding to a content of about 45 gms. diamine per litre.
168 gms. Dinitro-benzene.
300 gms, Fe.
20 c.cs. Hc1 (30 %).
The boiling solution is now treated very carefully with solid calcined soda in small portions (about 10 gms.). As soon as the reaction with litmus is distinct the product is boiled for a further 5 minutes, in order to decompose completely the soluble iron compound of any hydroxylamine which may be present. The liquid should not be filtered until a test on filter paper gives no black stain with sodium sulphide solution (1-10); This test should never be omitted on the large scale, and is likely to save much annoyance.1 The liquid is then filtered into a bolt-head which has been previously warmed, and the clear filtrate is treated with sufficient hydrochloric acid to cause a slight acid reaction to litmus. A solution of m-phenylene-diamine obtained in this manner keeps well. Yield about 95 gms., 100 %. The quantitative estimation is carried out in very dilute solution at o° with diazotized aniline, as in the case of H-acid, except that it is unnecessary to add soda.
This technical solution suffices for many purposes, but a purer diamine is preferable as the final yields are thereby improved. For this purpose the aqueous solution is evaporated first over a naked flame, and then preferably in vacuo, until it contains about 40 % of base; it may now be distilled in vacuo or, more simply, it can be frozen out at o°. In order to start the crystallization with this "cold " process it is necessary to "seed" the solution with a crystal of pheny-lenediamine. The purified diamine forms beautiful white prisms containing half a molecule of water, and differs from the impure product in being perfectly stable; 0- and p-diamines are present in the aqueous mother-liquor in concentrated form, which is the reason why the commercial liquor is so easily oxidizable. Orthodiamine gives immediately the characteristic blue-black coloration with aniline, acetic acid, and a little bichromate, by means of which it can be readily identified in the solution.
ca. 10 gms. Na2Co3.
1 If in spite of long boiling the iron reaction persists, the iron may be precipitated with a little ammonium sulphide.
The English firm of Read,Holliday & Sons 1 places a m-phenylene-diamine on the market, which has been recrystallized from water, and in spite of its somewhat higher price, it is strongly to be recommended owing to the excellent yields of colour obtained by its use.
The homologous 1:2:4-toluylene-diamine is obtained in a precisely similar manner to m-phenylene-diamine. It is characterized by its great purity even in its aqueous solution, as the technical dinitro-toluene is almost chemically pure; in spite of this, however, it is usually evaporated down and recrystallized in order to obtain a 100 % product. Dinitrochlorbenzene, p-nitraniline, and other insoluble nitro compounds behave in a similar manner.
The exceptions, however, are all those compounds containing carboxyl groups (Cooh). These cannot be reduced with iron in neutral solution, or only if special precautions be taken, as insoluble iron compounds are immediately formed. For instance, the important nitro-salicylic acid must be reduced by means of tin and hydrochloric acid, the tin being recovered without loss by precipitation with zinc dust. Chlornitro-benzoic acid is best reduced in neutral solution with zinc dust. Nitrophenol sulphonic acids, etc., however, are reduced by means of sodium sulphide (or hydro-sulphide) solution under pressure at 120-1500.
p-Nitraniline and p-Amino-acetanilide from Aniline,2
Reaction:

Aniline.
Acetanihde.
Nitro-acetanihde.
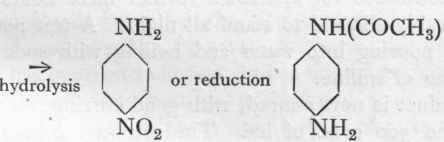
p-Nitraniline.
p -Amino-acetanilide.
1 This firm is now absorbed in the British Dyestuffs Corporation, Ltd.- F. A. M.
2 See also, P. Miiller, Ch. Z. 1912, 1049, 1055.
Continue to:


