Phenylhydrazine Sulphonic Acid From Sulphanilic Acid
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Phenylhydrazine Sulphonic Acid From Sulphanilic Acid
Reaction:

3/10 Gm.-molecule technical sulphanilic acid (100 %) is dissolved in 200 c.cs. water with the aid of 16 gms. of soda, the residual aniline being boiled off with steam. The filtered solution is cooled down in a glass vessel, 35 gms. conc, sulphuric acid are added, and the whole cooled further to 120 (external cooling); it is then treated with a solution of 21 gms. 100 % sodium nitrite in 50 c.cs. water (3/10 gm.-molecule) during half an hour, with continuous stirring, until a distinct and permanent reaction is given with nitrite paper. The diazosulphanilic acid comes out as fine crystals, which are filtered off at 12-140 on a small suction filter. The residue of crystals are rinsed out of the beaker by means of the mother-liquor.1
The diazo compound so produced is added to a mixture of 130 gms. sodium bisulphite solution (containing 25 % So2), and just enough 35 % caustic soda solution to cause the sulphite to give a distinct reaction with phenolphthalein paper; according to the quality of the technical bisulphite 25-45 gms. soda-lye will be required. If too little be used the hydrazine sulphonic acid becomes discoloured later on and deposits resinous matter. [The use of solid commercial sodium sulphite is unsatisfactory, as its So2 content is too variable.] The temperature of the mixture is kept below 500 by placing the vessel in ice water and stirring well. The diazo
51 gms.
Sulphanilic acid.
35 gms. H2So4.
21 gms. NaNo2.
130 gms. NaHso1 solution.
25-40 gms. NaOH (35 %)•
1 The moist diazosulphanilic acid is quite harmless, but it is highly explosive in the dry state, so that great care must be taken, sulphanilic acid is at once converted into the sulphophenyl-azo-sulphonic acid, which is allowed to stand for an hour.
It may be converted into the hydrazine sulphonic acid in various ways. The simplest is the following, which is also that used technically. The yellow solution of the azo-sulphonic acid is heated to boiling in a porcelain dish, and the boiling solution treated with 30 % hydrochloric acid (250 gms.) until the reaction is very strongly mineral acid. This operation should take about half an hour, in order to allow the sulphurous acid set free from the neutral sulphite sufficient time to exert its reducing action (fume cupboard !). If the solution should not by then have become decolorized a little more zinc dust may be carefully added. The phenylhydrazine sulphonic acid crystallizes out on cooling and, after standing 12 hours, is filtered off and washed with a little water. Yield about 50 gms. 100 % product.
The quantitative estimation is carried out by condensing a moderate amount of the substance with aceto-acetic ester in acetic acid solution and weighing the resultant pyrazolone.
Notes on Works Technique and Practice. - The preparation of aryl-hydrazine sulphonic acids has recently gained considerably in importance, as they are the starting-points for various yellow and red colouring matters which are fast to light, certain of which are mentioned later. Most amines of the benzene series may be similarly converted into the corresponding hydrazines; the chlor-phenyl-hydrazine sulphonic acids and the tolyl-hydrazine sulphonic acids, for instance, are largely manufactured. Certain of them are somewhat difficult to reduce completely, so that the elimination of the sulphonic group attached to the nitrogen atom only takes place at 110-135o. Such sulphonic acids, therefore, are either brought to the necessary temperature in lead-lined autoclaves, or else the difficulty is got over by the following neat device: sulphuric acid is used in place of hydrochloric acid for setting free the sulphurous acid, and is allowed to run under the surface of the boiling liquid. The reaction liquid thereby heats up very strongly and the sulphuric acid hydrolyses the sulphohydrazine sulphonic acid without difficulty; very occasionally it is necessary to add a little stannous chloride or zinc dust. On the large scale, with 1/2 kg.-molecule charges, the hydrolysis and reduction occupy about 3 hours. The yields are up to 95 %, and the crystallizations, as in the case of benzidine disulphonic acid, occupy several days.
ca. 250 gms. 30 % Hc1.
m-Nitraniline from m-Dinitro-benzene.
Reaction:
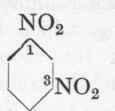
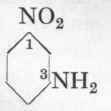
500 C.cs. of water are heated to 850 in an iron or glass beaker of 1 1/2 litres capacity, 100 gms. dinitro-benzene are then added, and by means of very efficient stirring are practically emulsified (propeller stirrer; caution required owing to the highly poisonous vapours). 245 Gms. of crystallized sodium sulphide (Na2S + 9 aq.) dissolved in 200 c.cs. of water are then allowed to run into this emulsion from a dropping funnel during 10 minutes. The dinitro-benzene is partially reduced by alkali sulphide, m-nitraniline being formed. The end-point of the reaction may be recognized by the fact that a drop of the solution on a filter paper gives a black streak of metallic sulphide with an iron or copper sulphate solution. As soon as the blackening remains for 20 seconds, the mixture is cooled at once to 200 by throwing in 500 gms. of ice, and, after standing for several hours the m-nitraniline is filtered off; it may be recrystallized from boiling water, but for most purposes this is unnecessary. For works use the crude product is simply dissolved in hydrochloric acid, the sulphur filtered off, and the solution utilized directly. The yield is about 55 gms. pure recrystallized m-nitraniline.
A solution of sulphur and sodium sulphide has also been recommended for the reduction, but this process is not advised, as it leads to poorer yields and less pure products.
It may be noted here that certain nitro compounds cannot be partially reduced in this simple manner, as the reduction for the most part goes too far; for further details, see the reduction of picric acid to picramic acid (p. 77).
Continue to:


