Hard Soldering
Description
This section is from the book "Art Metalwork With Inexpensive Equipment", by Arthur F. Payne. Also available from Amazon: Art Metalwork with Inexpensive Equipment.
Hard Soldering
The handles and spouts on such objects as kettles, tea-pots, and pitchers, are usually soldered into place with hard silver solder, and should seldom be riveted, or soldered with soft solder. There are many formulas for making hard or silver solder, but that which is best for our purpose is composed approximately of silver 8 parts, copper 3 parts, spelter (zinc) 1 part. This makes an easy flowing solder for copper and brass. A fair solder for this work may be made by melting together 2 parts of silver and 1 part of high brass. Brass is made of copper and zinc, high brass having a larger percentage of zinc than low brass. Melt together on a piece of charcoal or asbestos, it will run into a ball, and while it is still melted quickly place a flat piece of iron on it; this will flatten it out so that it may be cut into small pieces more readily.
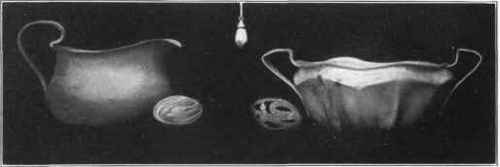
Fig. 101. Silver pitcher and sugar bowl, with forged wire handles.
The place where the handle or spout is to be soldered on must always be planished before the handle is tied on, because it would be very difficult to planish it after the handle is soldered on.
Hard soldering on copper and brass is easily done if these three points are observed: The joint to be soldered must be filed clean; every part of the joint and every bit of the solder must have borax on it; the work must be made red hot to cause the solder to run into the joint. The borax mentioned is the powdered borax that can be obtained at any drug store. It must be mixed with water to the consistency of cream and applied to the joint with a small brush. The handle is tied on with fine iron wire, Fig. 94. The solder is cut into very small chips and placed at the joint and the heat is applied slowly until the borax is dry; then apply the full power of the blow-pipe until the solder melts and runs into the joint.
To clean the pitcher and to remove the melted borax place it for about thirty minutes in a "pickle" made up of one part sulphuric acid and two parts water. Clean it with the wire brush or emery cloth, planish smoothly all over, then color and wax as described before.

Fig. 102. Blow-pipe. No. 8c.
Fig. 103. Blow-pipe. No. 8e.
For hard soldering, annealing, and melting such as we would do in making the problems in the series, there is nothing better than the 8C blow-pipe for small work, Fig. 102, and the 8E blow-pipe for the larger work, Fig. 103. Both blow-pipes are to be used in combination with the 10A foot blower, Fig. 104. If power is convenient, and more than one blow-pipe is to be operated at one time, a power blower is best. If no gas is available, a 40c gas generator is very good, or a plumber's blow-torch may be used for small work. If a large soft flame is desired an O-L No. 82 blow-torch is best and for a sharp needle flame O-L No. 29.

Fig. 104. 10A Foot blower.
Figs. 105, 106, and 107 show a number of solid silver cream pitchers, sugar-bowls, and salt-dishes. While most of the problems described in this series have been made of copper and brass, the instructions given would be the same for making the same objects in silver, with this important exception, that the annealing of silver should be done in a rather dark place so that it can easily be seen when it becomes red hot, and it should never be heated to more than a dull red or it will be very liable to melt. Silver melts at 1830 degrees Fahrenheit, and copper at 1995 degrees, so it will be seen that the melting point of silver is considerably lower than that of copper.

Fig. 105. Solid silver pitcher, one-piece hollow handle.
From Kalo Shop, Chicago.
The feet on the salt-dishes were made by melting scrap silver into little balls on a charcoal block; it is a peculiarity of metals that if a small quantity is melted on a flat surface, it will run together in the form of a ball. Three balls of equal size were made and soldered with the silver solder to the bottoms of the dishes, thus forming the feet.
Silver articles may be oxidized with the same sulphide of potassium solution that was suggested for use on copper. See p. 41.

Fig. 106. "Paneled" silver salt dishes.
To etch a design on silver proceed as with copper, except that the etching solution is made of nitric acid, 20 parts; hydrochloric acid, 5 parts; water, 75 parts. Or, a solution of equal parts of nitric acid and water may be used.

Fig. 107. Silver sugar-bowls, fluted and paneled, wire handles.
Articles made of silver may be polished by hand by first using fine emery cloth. Then mix a paste of flour emery or powdered pumice with oil, and apply with the piece of emery cloth. Nearly all metals under certain conditions will form a combination with the oxygen in the air. Iron rusts, forming oxide of iron; copper when heated red hot forms a black scale which peels off when it gets cold, this is black oxide of copper. Silver when it is heated to redness also forms a thin white scale (designated in the trade as "fire scale"), that clings tenaciously to it. In fact, whenever it is desired to remove it, it is necessary to file, scrape, or polish it off, or else remove it with acid. The reasons for mentioning it here are: First, when soldering two pieces of silver together, both pieces should always be filed so as to cut thru this fire scale, otherwise they are very liable to break apart; Second, when polishing a finished piece of silver one is very liable to polish thru the fire scale, and as the fire scale is of a different shade of color from the silver underneath, it makes the object look patchy in color. So it is necessary either to have all the fire scale off, or else have it all on. The old craftsmen left it all on. The modern manufacturers take it all off by dipping it in a solution composed of equal parts of nitric acid and water, to which is added a small quantity of acetic acid (one-half pint acetic acid to two gallons of the compound) to make it work smoothly. For our work it is just as well and very much easier to leave it all on, so after the silver has been well polished and all the scratches and marks removed, heat it with the blow-pipe to a dull red, and when it is nearly cold place it in the sulphuric acid "pickle" and leave it there for ten minutes, then repeat the process, and the silver will be a dull white (care must be taken not to put any iron or steel in the pickle, as it will turn the silver copper color). Then it should be polished lightly with the emery paste, washed off in hot water with soap, and finally polished with canton flannel and powdered rouge or whiting.

Fig. 108. Silver salad-bowl, with two-piece handle.
From Kalo Shop, Chicago.
This treatment will give to the silver the soft, dull sheen that has added so much to the charm of the old colonial silver, and which is so much more beautiful than the hard, bright glitter of the modern commercial finishes.
The aluminum bread-tray, Fig. 109, was beaten up from a flat piece of metal by methods similar to those described for working copper, except that it is not necessary to anneal aluminum.
Aluminum can be cleaned by immersion in a solution of caustic soda.

Fig. 109. Aluminum bread-tray.
Continue to:


