Refining Copper Metal
Description
This section is from the book "Modern Shop Practice", by Howard Monroe Raymond. Also available from Amazon: Modern Shop Practice.
Refining Copper Metal
Furnace Method. Practical Necessity
The great bulk of copper as produced in converters and as turned out is by no means pure enough for refining by electricity; in fact the electrolytic refining of copper is possible commercially only when the metal to be refined already is of very high copper content. Because of this, if the blister copper is not melted in a reverberatory at the smelting plant, it necessarily is done at the copper refinery, which may be located at some point better situated for the obtaining of cheap power - the most essential feature of electrolytic refining.
The furnace refining of nearly pure copper is, then, an essential step in the production of merchantable material. It is carried out extensively at all copper refineries, both on blister copper and on the cathodes produced by electrolytic refining. The different sorts of blister copper are susceptible to extensive furnace refining, but the melted cathodes require it in slight degree only and that largely for the exact regulation of the final oxygen content of the metal.
Partial Separation Of Metals
Furnace refining can almost completely remove iron, lead, tin, sulphur, manganese, and zinc; arsenic and antimony are partly removed by this refining, but it will have little affect on selenium, tellurium, bismuth, or the precious metals. This accounts for the fact that furnace refining is an essential operation, yet is unable to produce a commercially pure copper as demanded in the trade.
Process. Oxidation
This furnace refining of copper consists in melting down the copper in a large coal-fire reverberatory, during which melting considerable of the impurities will be oxidized and floated on the surface of the metal. More extensive oxidation can be effected by flapping the metal with iron paddles or by blowing in air through iron pipes. This process of oxidation, however, introduces undue amounts of oxygen, which is absorbed by the metal and retained in solution. The oxidized slag having been removed, the excess of oxygen then can be taken out of the metal by covering with charcoal and thrusting in logs of wood, which, decomposing in the hot bath, use up and remove the oxygen.
Electrolytic Method. Usage
Nearly all crude copper is electrolytically refined; this means that after the metal has been smelted to black copper, blister copper, or any sort of crude cakes, they are remelted and somewhat purified in an anode melting furnace and cast into anodes. The anodes are refined in the electrolytic cells and the cathodes melted, before their final exit from the refinery as ingots, slabs, and wire bars.
This type of refining is applied to most of the metal won by the reducing methods referred to previously - the main exception is some of the Lake Superior copper which is refined in the smelting furnaces, as it was originally quite pure.
Complete Separation Effected
From a reasonably pure anode, as results from the remelting of blister copper in the refining furnace, electrolytic refining is able to produce a metal of the most extreme purity, except in that particular element, oxygen, which again will be introduced in the final remelting as will be necessary to make shapes, ingots, slabs, and wire bars.
The trade demands at the present time are extremely exacting, and most of the copper of commerce, therefore, is put through this process of electrolytic refining. Further than this, electrolytic refining effects an extraordinarily complete separation of the precious metals, the removal of which it is equally difficult to accomplish by any furnace process.
Process. Arrangement Of Electrodes
Electrolytic refining consists of dissolving copper from an anode immersed in a strongly acid solution of copper sulphate, of forcing the electropositive particles of copper through the solution, and of plating them out on a sheet of pure copper suspended close to the anode; the particles coming out on this near-hanging strip of copper will constitute the cathode; the force accomplishing this transfer is of course the electricity which is supplied to these thousands of couples in great quantity by the generating system in the refinery. It is common to hang about twenty of these couples in an acid-proof tank, which is about 3 feet wide, 4 feet deep, and 10 feet long. The electric current is sent through each tank, with the respective anodes and cathodes arranged in parallel. A number of tanks will be put in series and arranged in groups to accomodate the amperage and voltage most suitable as generated by the large dynamo units.
Action Of Electrolyte
The electrolyte is kept at a very definite concentration of copper and free acid and is maintained in active circulation through the vats at a constant temperature of between 60° and 70° C. The electrolytes of different plants average close to 4 per cent of copper and 12 per cent of free acid. Anodes waste away rapidly due to the solution of the copper and in about a month's time are taken out and new ones substituted. The cathodes are taken out more frequently, when the freshly deposited copper is stripped from the starting sheets, and the sheets are put hack again for a new layer.
The anodes which are taken out of the vats are washed to remove the slime and then are remelted into full-sized anodes to be put again into the tanks. Fig. 38 indicates a series of these anodes as lifted out from one of the tanks, which are seen together in the other view thickly packed on the floor of the large electrolyzing room. Separation of Dore Metal. A large quantity of sediment collects in the bottom of the electrolyzing vats and is washed off from the anodes when they are cleaned. This mud is carefully removed from the vats and collected by itself in large tanks for further treatment, t contains the impurities originally present in the anode, of which the most important are gold, silver, palladium, and platinum. Selenium and tellurium also are undesirable components of this mud. This mud is washed, its copper content largely depleted with a sulphuric-acid treatment, the selenium and tellurium oxidized away in a small reverberatory, and a metal known as dore metal finally obtained which goes to the silver refinery for separation and recovery of the gold, silver, palladium, and platinum.
Fig. 38. Electrolytic Vats and Anaconda Courtesy of Engineering and Mining Journal".
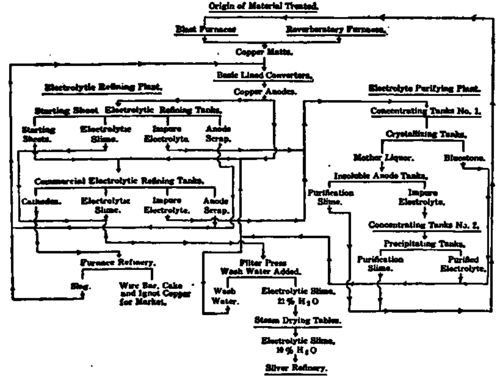
Fig. 39. Flow Sheet of Copper Refinery Courtesy of American Institute of Mining Engineers.
Diagrammatic Summary
Fig. 39 may be studied to advantage as a summary of the course of materials and processes in a refinery. It is necessary that the diagram take up the material as produced by blast furnaces or reverberatory furnaces and that it turn out market copper or partly dried slimes to go to the silver refinery. The main electrolytic treatment for the bulk of the material is on the left-hand side of the diagram; the right-hand side is devoted to the course pursued in purifying the electrolyte, a small portion of which is being separated continuously and treated to get out the accumulating impurities. This diagram may be studied in every detail to much advantage.
Continue to:


