Lime
Description
This section is from the book "The Profession Of Home Making", by American School Of Home Economics. Also available from Amazon: The Profession Of Home Making.
Lime
One of the common chemical substances found about the country house at least is quick lime, used for whitewash and as a deodorizer.
The term lime usually means the oxide of the element calcium. Its commonest compound is calcium carbonate which is found in nature as limestone, chalk, marble, coral, shells, and several other familiar substances. Calcium is also found combined with sulphur and oxygen in the compound calcium sulphate, which is the mineral gypsum from which plaster of Paris is made. Bones contain a considerable amount of calcium phosphate and egg shells, calcium carbonate.
Lime, the oxide of calcium, is made by heating broken pieces of limestone in furnaces called lime kilns. The calcium carbonate as a compound is broken up, carbon dioxide gas being given off and calcium oxide left. This freshly formed oxide is called "quick lime," and when it is exposed to moist air, it attracts water and changes to a form called chemically, calcium hydroxide and, commonly, "slaked lime." Quick lime may be used to dry the air of damp cellars, etc., because of this property. The process of slaking the lime is also accomplished by treating quick lime with water. When this is done, much heat is evolved and the hard lumps crumble to a soft powder and increase considerably in bulk. The rise in temperature shows that chemical change is taking place.
Slaked lime will dissolve slightly in water, yielding lime-water. This is a mild alkali and has several household uses. It may be prepared by pouring two quarts of boiling water over about a cubic inch of unslaked lime. Stir it thoroughly and let it stand over night; in the morning pour off the liquid and treat the sediment with hot water a second time. When the sediment has again settled, pour off the clear liquid and bottle this. It is mixed with milk and fed to young children and invalids to prevent acidity of the stomach and make the milk more easily digested. Lime-water and oil form one of the best remedies for burns. The alkali of the lime neutralizes the acid nature of the burn.
Mortar is made of slaked lime and sand. When this is spread upon the walls, the lime slowly absorbs carbon dioxide, always present in the air, and changes to carbonate of lime. The water is given off into the air (evaporates) and the mass becomes hard. Of course the surface becomes carbonate sooner than the deeper parts because this has closer contact with the air, and it therefore takes considerable time for all the plaster to harden. The water contained in the mortar soon dries, but while the mortar is becoming hard, more water is continually formed in the chemical process, so that it requires a long time for the new plaster to become quite dry. It is considered unhealthy to live in rooms with newly plastered walls. This may be because such walls are damp, thus producing damp air, or it may be because the moisture in the walls interferes with the passage of air and other gases through the walls - a process little considered as a rule, but of great importance.
Certain varieties of limestone contain other salts, such as magnesium carbonate. Lime made from these does not soften from exposure to the air. It will, however, harden after long contact with water, and such substances are known as cements. Portland cement will harden under water.
Mortar and
Plaster
Fig. 24. Acetylene Gas Burners
Hydraulic Cement
Quick-lime is a strong alkali and does the work of such substances. It is used in tanneries in taking hair from hides and also in decomposing fats for making candles. When dead animal substance is buried in lime, the process of decomposition is greatly hastened, probably because the lime unites with all water present while the strong alkali acts upon the fats reducing them to soaps of different kinds.
Whitewash is simple slaked lime mixed with water. It is very cleansing in its effects and also gives the appearance of freshness and cleanness. When newly applied, it is nearly colorless, for the calcium hydrate is colorless; this in the air soon changes to calcium carbonate which is white and opaque.
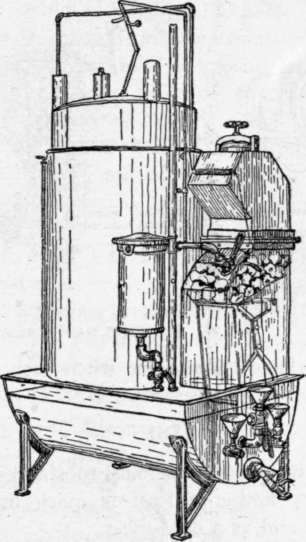
Fig. 25. Acetylene Gas Generator and Storage Tank
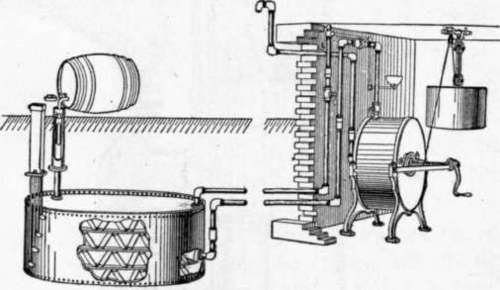
Fig. 26. Gasoline Gas Plant
Continue to:


