A Study Of Baking-Soda And Baking-Powder
Description
This section is from the book "Elements Of The Theory And Practice Of Cookery", by Mary E. Williams. Also available from Amazon: Elements Of The Theory And Practice Of Cookery; A Textbook Of Domestic Science For Use In Schools.
A Study Of Baking-Soda And Baking-Powder
Experiments
A. Dissolve half a teaspoonful of baking-powder in about two tablespoonfuls of water. What happens? B. When the solution stops bubbling (effervescing) heat it (in a saucepan on the stove or in test-tube over a bunsen burner or a gas-stove burner). What effect has heat on the bubbling? C. While it is bubbling fast, hold a lighted match over the mouth of the test-tube. What gas is being formed? This gas may also be tested for by the apparatus shown in Fig. 7. What effect will the gas have on the lime-water (p. 6) ?
How Baking-Powder Makes Biscuit Light
When baking-powder is dissolved, carbon dioxide is formed and bubbles up. Heat increases the action. When the action takes place in a mixture during cooking, the thousands of bubbles of gas formed are caught in the mixture and baked in. This makes the biscuit, muffins, or cakes porous and light.
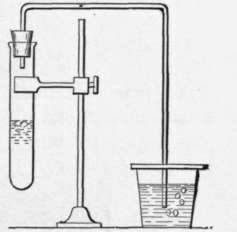
Fig. 7. - Apparatus for testing for carbon dioxide.
Test-tube (t) containing baking-powder solution. Glass tube connected with t dips into lime-water contained in glass (0).
Experiments
D. Dissolve a teaspoonful of soda in a fourth of a cup of water in one glass, and two teaspoonfuls of cream of tartar1 in the same quantity of water in another glass. (Cream of tartar dissolves more readily in hot water than in cold.) E. Pour the two solutions together. Observe the effect and tell what you think causes it. F. If a further demonstration is desired, a little soda with about twice as much cream of tartar may be dissolved, heated in a test-tube, and the gas tested as in Exp. B. G. Add a pinch of soda or a little soda solution to sour milk; to vinegar. H. Taste cream of tartar. How does it taste?
How Soda Makes Griddle Cakes Light
Soda is a carbonate. When such a carbonate as soda and an acid meet in solution, they unite, and carbon dioxide is formed. In sour-milk griddle cakes the sour milk supplies lactic acid.
Baking-powder contains both soda and cream of tartar or some other acid.
Experiments
I. Dip a strip of red litmus2 paper into a soda solution. Dip a strip of blue litmus into a cream of tartar solution. Test with litmus paper sour milk, vinegar, washing-soda, soapy water, ammonia, a salt-solution, and a solution of baking-powder. Which of these sub-stances turn red litmus blue? Which turn blue litmus red? Which do not alter the color? J. Taste griddle-cake batter. Test it with litmus.
Acids, Alkalies, Salts
Any substance that turns blue litmus red is an acid. Any substance that turns red litmus blue is an alkali.1 A substance that does not affect the color of litmus is said to be neutral.2 The union of any-acid and any alkali used in cooking produces not only-carbon dioxide, but a neutral salt. (See p. 57.)
1 Cream of tartar is a product of grapes. It is made by purifying the crystals, called "argols," which form in wine-vats.
2 Litmus is a coloring matter made from lichens which grow on the coasts of Europe.
Why Sour-Milk Griddle Cakes Are Not Sour
This explains why griddle cakes, although made from sour milk, do not taste sour. If just enough soda is used to neutralize the lactic acid, the batter will not change the color of litmus. Usually, however, more than enough soda is used, and the batter turns red litmus blue. The salt formed by the action of lactic acid on soda is harmless. One teaspoonful of soda is usually allowed to one pint of sour milk.
Different Kinds Of Baking-Powder
A baking-powder which contains just the right proportions of soda and acid makes a neutral solution. It is important that the salt formed should be harmless. Cream of tartar forms with soda Rochelle salts. These are soluble. They have a slight medicinal effect, but in eating food raised with baking-powder, a person gets a very small quantity of the salt. Cream of tartar baking-powders, also called tartrate powders, are among the best on the market. Two other classes of baking-powders are made. In one of these the acid used is acid phosphate. In the other it is alum. The salt left in food raised with phosphate powders is insoluble, hut not injurious. Alum powders leave an objectionable residue. Alum powders are cheap, but as they lose their strength sooner than tartrate powders do, and as they often contain considerable starch, an alum powder may not be as cheap as it seems.
1 Note that alkaline solutions feel slippery. Washing-soda and baking-soda are both alkaline carbonates. Washing-soda is sodium carbonate, baking-soda is sodium bi-carbonate.
2 Water in which red cabbage-water has been boiled may be used instead of litmus paper as a test for acids and alkalies. Acids turn cabbage-water violet. Alkalies turn it green.
Starch In Baking-Powder
All baking-powders contain some starch, put in to absorb moisture. If the acid and soda are not kept dry, they begin to act, and some of the carbon dioxide is lost. If more starch than is necessary for this purpose is used, it is an adulteration. (See p. 53.) Baking-powder may be tested for starch (a) by boiling and (6) with iodine (p. 61).
To keep baking-powder dry, always cover the box as soon as you have taken out what you need.
Proportion Of Baking-Powder To Flour
Use one to one and one-half teaspoonfuls of baking-powder to one cupful of flour. If more than this is required, the baking-powder is of poor quality. Bread or cake made with " generous " measures of baking-powder is dry, and contains an excess of Rochelle salts. One scant teaspoonful of soda and two level teaspoonfuls of cream of tartar are equal to three teaspoonfuls of good baking-powder. It is better to use a good baking-powder than to use soda and cream of tartar, because the housekeeper cannot proportion the two as accurately as the manufacturing chemist. Any excess of either is wasted, and may be injurious to health.
How The Carbon Dioxide Raises The Batter Or Dough
As cream of tartar is only partly soluble without heat, little of the gas is set free until the mixture is put into the oven.
It then comes off rapidly, filling the batter or dough with bubbles, and making it rise higher and higher. As the gas expands, the walls of the bubbles stretch and become thin. Just at this stage, if the oven is right, the heat sets the mixture and imprisons the gas. In too hot an oven a crust forms before all the gas is set free; in too cool an oven the bubbles break and the gas escapes. In either case the result is heavy bread.
Continue to:


