165. Eucalyptus dives
Description
This section is from the book "A Research On The Eucalypts Especially In Regard To Their Essential Oils", by Richard T. Baker, Henry G. Smith. Also available from Amazon: A Research On The Eucalypts And Their Essential Oils.
165. Eucalyptus dives
(Schauer, in Walp. Rep., ii, 926.) Peppermint or Broad-leaved Peppermint.
Systematic. - A tree often attaining very large dimensions, although it is found in flower and fruit when only a few feet high (Mittagong Ranges, Berrima Coal Mine, Fagan's Creek, Braidwood District), and when its leaves are sessile, cordate. Bark greyish, furrowed, and somewhat similar in appearance to that of the "Messmate" (E. phellandra), but yet of a stringy nature. Abnormal eaves ovate cordate sessile, measuring several inches in length; venat on marked, intramarginal vein removed from the edge Normal leaves lanceolate, petiolate; venation well marked on both sides, very oblique, intramarginal vein removed from the edge. Flowers numerous, up to twenty, on axillary peduncles under 1 inch long.
Fruit. - Varying between pear-shaped and hemispherical, shining; rim thick; valves not exserted or only slightly so; 3 lines in diameter.
These somewhat resemble E. amygdalina and allied species, and the smaller forms of E. vitrea.
Habitat. - Common throughout the whole coastal ranges of New South Wales and Victoria.

Plate LXXV.

Eucalyptus Dives. Sch. A Broad Leaf Peppermint
Plate LXXVI.
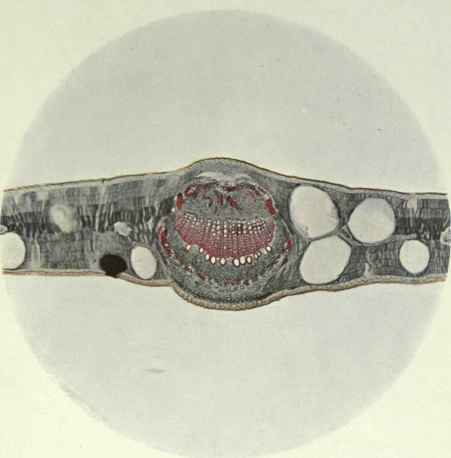
This species which yields so large a quantity of oil, was naturally expected to have large and numerous oil glands as are shown in the plate. The palisade tissue is of less area on the upper than the lower side of the mesophyll, which, in this case stands out more distinctly than in most sections illustrated. The cuticle shows no variation on either side of the leaf. The mid-rib in the centre of the plate illustrates the typical bicollateral structure of the vascular system of the Eucalypts, the xylem being bordered top and bottom by phloem. Towards the upper side of the xylem are the elements of the protoxylcm, lower down are the smaller xylem fibres, and lower again the larger vessels. Outside the phloem are the thick-walled strengthening fibres and further layers of supporting tissue. The black spot on the left of mid-rib is a fungus. x 55.
Eucalyptus Dives, Schau
REMARKS, Both Bentham and Woolls were in accord with Schauer in regarding this tree as a distinct species, but Mueller synonymised it with E. amygdalina, Labill, and this has caused the latter species to lose much of its good name concerning its economics, as, tor instance, its oil. In a natural classification the two species never can be confounded. The leaves of E. dives are very variable in shape and size, being either sessile, opposite and cordate or ovate-lanceolate on the same tree, It also Bowers and fruits in a very early stage of its growth (5 to 6 feet), when the peduncles a1 the base of the cordate, sessile leaves, differentiate it from E. amygdalina, whilst in this latter feature it resembles the "Argyle Apple" (E. cinerea, E.v.M.) or E. pulverulenta. The early foliage is also quite different from that of B. amygdalina, not to mention the nature of the bark, timber, and constituents of the oil. It is, therefore, all the more difficult to understand now, on morphological grounds, the two should have been thought to be one and the same species. It is quite a distinct tree.
ESSENTIAL OIL. - Leaves and terminal branchlets for distillation were obtained from Fagan's Creek, Braidwood, N.S.W., in October, 1898. The yield of oil was 1.96 per cent. The crude oil was almost colourless, and had a strong peppermint odour, due to the large amount of piperitone it contained. It consisted very largely of phellandrene. Cineo did not exceed 5 to 8 per cent, in the crude oil. The third fraction consisted largely of the peppermint ketone, piperitone.
The crude oil had specific gravity at 150 C. = 0.882; rotation aD - 63.9°; refractive index at 200 = 1.4837, and was soluble in 2 volumes 80 per cent, alcohol The saponification number for the esters and free acid was 2.9
On rectification, 2 per cent, distilled below 1720 C. (corr.). Between 172-198°, 60 per cent. distilled; between 198-227°, 13 per cent came over, and between 227-240°, 20 per cent. distilled. These fractions gave the following results: -
First fraction, sp. gr. at 150 C. | = | 0.8593; | rotation aD | - | 73.8°. | |||
Second | " | " | " | = | 0.8936; | " | not taken. | |
Third | " | " | " | = | 0.9318; | " aD | - | 9.4°. |
Material of this species for distillation was also obtained from Barber's Creek, N.S.W., in October, 1898. The yield of oil was 2.1 per cent.; the crude oil differed in no respect from the above sample, and this result is a good illustration of the comparative constancy of constituents in the oil of the same species at the same time of the year. The specific gravity of the crude oil was 0.882, and optical rotation aD - 63.6°. The crude oil formed a clear solution with 1 volume 80 per cent. alcohol, and the peppermint ketone, piperitone, was present in great quantity in this sample.
Material of this species for distillation was also obtained from Berrima, N.S.W., in May, 1899. The yield of oil was 2.9 per cent. The specific gravity of the crude oil was 0.8887, and optical rotation aD - 55.8°. It was in agreement in all respects with those mentioned above, and formed a clear solution with 1 volume 80 per cent. alcohol.
Material of this species was also obtained from Rylstone, N.S.W., in August, 1898. The oil was practically identical with the above.'
During recent years, considerable quantities of the oil of this species have been distilled in Australia. It is the best of all essential oils for use in the Mining Industry for the separation of mineral sulphides by a flotation process, and as the yield is large it can be produced somewhat cheaply. Previously the oil of E. dives was forwarded to Europe as the product of E. amygdalina; that this is so can be seen from the study of the constants recorded in the older literature.
The oil of E. dives shows a comparative constancy similar to those of other individual Eucalypts, so that the species has now become stabilised.
This subject is more fully treated in the article "The comparative constancy of the oil products from individual species of Eucalyptus,"
The cineol in the oil of this species is always small in amount, averaging probably between 5 and 8 per cent. It is impossible to determine this small amount accurately by the phosphoric acid method, or by any other known method besides absorption with 50 per cent, resorcinol. Statements have been made that the resorcinol method indicates the presence of a much larger quantity of cineol than the oil actually contains, and that is so, if the results of the direct absorption be taken without correction. On rectifying the oil so as to separate the constituents boiling between 170-190° C, a considerable quantity of piperi-tone comes over, and is absorbed by the resorcinol at the same time as the cineol. The piperitone can, however, be readily estimated by a ketone determination, using the normal sodium sulphite method, and the amount so found when subtracted from the total absorbed, will give the cineol.
An actual determination will make this clear. A sample of freshly-distilled oil of E. dives was forwarded to us by Mr. A. J. Bedwell, in November, 1919; it was typical of the oil of this species. The crude oil had: -
Specific gravity at 150 C. ................... | = | 0.8828. |
Optical rotation aD................... | - | 68.0° |
Refractive index at 200 ...................... | = | 1.4769. |
Soluble in 1 volume 80 per cent, alcohol. Ketone determination................... | = | 29 per cent. |
65 per cent, distilled below 190 ° C. (corr.). This had: - | ||
Specific gravity at 150................... | = | 0.8592. |
Rotation aD ..................... | - | 77.2°. |
Refractive index-.at 20° ...................... | = | 1.4734. |
Resorcinol absorption................... | = | 22.5 per cent. |
Ketone determination................... | = | 10.0per cent. |
Cineol after correction................... | = | 12.5 per cent. |
When calculated for the original oil the result was 8 per cent. of cineol.
The portion boiling above 1900 C. (35 per cent. of original oil), had specific gravity at 150 C. = 0.9335; rotation aD - - 38.2°; refractive index at 200 C. = 1.4855; and ketone estimation 75 per cent.
The non-ketone portion of the original oil had specific gravity at 15° C. - 0.8633; rotation aD - 70.2°; refractive index at 200 = 1.4746; saponification number = 15.93, and after acetylation = 38.35, which calculated for a C10Hl8O alcohol gives 6.3 per cent. This alcohol is probably piperitol, largely, as in the oil of E. radiata.
Continue to:


