Limonene
Description
This section is from the book "The Volatile Oils Vol1", by E. Gildemeister. Also available from Amazon: The Volatile Oils.
Limonene
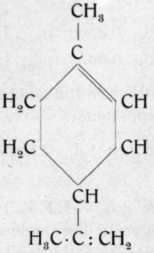
Limonene is a very widely distributed terpene. It is found in volatile oils, not only in the dextrogyrate and laevogyrate modifications, but also in the racemic modification, which is called dipentene and which is commonly treated as a separate hydrocarbon.
d-Limonene is contained in the oils of the Aurantiaceae, most abundantly in the oils of orange, lemon, bergamot and mandarin, in Italian and West Indian limetta oil, in neroli oil and petitgrain oil, also in Virginian cedarleaf oil, in the needle oil from Callitrisglauca, in gingergrass oil, in camphor oil, kuromoji oil, the oil from the fruits of Pittosporum undulatum, cedro oil, Manila elemi oil, oil of buchu leaves, caraway oil, dill oil, Macedonian fennel oil, celery oil, oil of Monarda punctata and erigeron oil.
/-Limonene has been found in the oils from the cones and leaves of Abies alba, in the turpentine oil of Pinus serotina, in Finland turpentine oil (?), in the oils of Monodora Myristica and Eucalyptus Staigeriana, in American peppermint oil, in Russian spearmint oil, in the oil of Bystropogon origanifolius, in American pennyroyal oil, in verbena oil, and in oil of goldenrod.
Limonene of unknown rotation has been found in the oils of rue and myrrh, in a fagara oil and in oil of Monarda fistulosa.
Synthetically, limonene has been prepared repeatedly.
By means of fractional distillation, (/-limonene can be obtained with a fair degree of purity from oil of orange or oil of caraway; /-limonene can be obtained in like manner from the oil of the cones of Abies alba. When carefuliy purified, the hydrocarbon has an agreeable lemon-like odor. The physical constants are approximately expressed by the following figures:
B. p. 175 to 176°; d15o 0,850; nD20o1,475.
For /-limonene from the leaves of Abies alba Wallach found:
B. p. 175 to 176°; d20o 0,846; nD20o1,47459.l)
The angle of rotation is high, but varies with the method of preparation. The highest deviation recorded was observed in the laboratory of Schimmel & Co. in connection with a d-limonene obtained from caraway oil by fractionation in vacuum, viz., [a]D + 123° 40'. A synthetic limonene prepared by Godlewski and Roshanowitsch2) gave [a]D -+- 125° 36'. For /-limonene from pine needle oil, Wallach3) records [a]D - 105° (in alcoholic or chloroformic solution). In connection with /-limonene from pine cone oil, the following constants were observed in the laboratory of Schimmel & Co.
B. p. 176,5°; d15o 0,8472; aD - 101° 10', [«]D - 119,41°; nD20o 1,47303.
Chemically both limonenes are alike, yielding the same derivatives which differ only in the direction in which they deviate polarized light. When d- and /-limonene are mixed, dipentene results. This also results when the active limonenes are heated to a higher temperature or when they are acted upon with acids. In the cold, acids frequently cause the hydration of limonene to terpineol and terpin hydrate. At elevated temperatures, however, acids dehydrate these same hydration-products with the reformation of hydrocarbons. Concentrated sulphuric acid changes limonene to cymene. When perfectly dry, limonene absorbs one molecule of hydrohalogen with the formation of limonene monochlorhydrate which, when treated with sodium acetate, yields active a-terpineol.4) A second molecule of hydrohalogen is added only in the presence of moisture when a derivative of dipentene (see p. 313) is formed.
1) Liebig's Annalen 246 (1888), 222.
2) Journ. russ. phys. chem. Ges. 31 (1899), 209. According to Chem. Zentralbl. 1899, I. 1241.
3) Liebig's Annalen 246 (1888), 222; Wallach and Conrady, Ibidem 252 (1889), 145.
4) Semmler, Berl. Berichte 28 (1895), 2190; Wallach, Liebig's Annalen 350 (1906), 154.
Limonene adds 4 atoms of bromine yielding an optically active limonene tetrabromide that melts at 104 to 105° (see below). Upon addition of nitrosyl chloride, each limonene yields two (a- and B-) nitrosochloridesl) which are physical isomers. These are prepared like pinene nitrosochloride and because of their different solubility in chloroform can be separated by means of this solvent. Chemically these nitrosochlorides behave alike. Upon withdrawal of hydrogen chloride with alcoholic potassa, they are converted into carvoxime melting at 72°. When treated with organic bases two series of nitrolamines2) result. Of these the a-nitrol piperidine base melts at 94°, the B-compound at 110°; the a-nitrol anilide base at 112°, the B-compound at 153°; the a-nitrol benzylamine base at 93°. The limonene monochlorhydrate mentioned above also yields a nitrosate.
With dilute permanganate solution limonene is oxidized to the saturated tetratomic alcohol limonetrite (m. p. 191,5 to 192°).3) The other oxidation products of limonene afford little that is characteristic.
Limonene is most readily identified by means of its tetra-bromide, prepared according to Wallach's directions.4) The fraction is purified as much as possible and diluted with about four times its volume of glacial acetic acid. Bromine is added drop by drop to the well cooled solution as long as it is taken up with decoloration. The crystals separating after standing for some time are collected and recrystallized from acetic ether. As a rule they are distinguished from those of dipentene tetrabromide by having smooth surfaces, and by being soft and fleible.5) The melting point of the pure tetrabromide is about 104,5°, the specific rotation of the chloroform solution is +73,27 and - 73,45° respectively.
In connection with the preparation of the tetrabromide, it should be noted that reagents absolutely free from water need not and should not be used. These are apt to give rise to the formation of a non-crystallizable tetrabromide. Nevertheless the crystalline product is to be regarded as the normal bromide.6)
1) Wallach, Liebig's Annalen 252 (1889), 109.
2) Wallach, Ibidem 270 (1892), 172.
3) Wagner, Berl. Berichte 23 (1890), 2315.
4) Liebig's Annalen 239 (1887), 3.
5) Wallach, Liebig's Annalen 227 (1885), 279. 6) Wallach, Ibidem 264 (1891), 14.
Wallach also advises not to use alcohol or ether as diluent since they give rise to the formation of liquid by-products. Like disturbing factors are encountered when the terpene fractions used are not sufficiently uniform in composition.
Minor modifications in the method of preparation have been recommended by various investigators. Baeyer and Villiger1) employ as diluent a mixture of 1 vol. of amylalcohol and 2 vol. ether, cool the mixture and add bromine drop by drop. In the proportion in which the ether evaporates, the tetrabromide is said to crystallize out.
Power and Kleber2) add the fraction to be tested for limonene drop by drop to a cooled mixture of glacial acetic acid and bromine until only a slight excess of bromine remains. The solution is then decolorized with an aqueous solution of sulphur dioxide and the bromide precipitated with water. Their object is to prevent the formation of hydrogen bromide and of noncrystalline bromides of isomeric terpenes.
Finally, a combination of both methods has been suggested by Godlewsky.3) He suggests that a solution of the terpene in a mixture of equal parts of amylalcohol and ether be added drop by drop to an ethereal solution of bromine which is kept cold by ice water during the course of the reaction.
Continue to:


