Carcinogenic Compounds And Chelating Agents
Description
This section is from the book "Chemistry Of Chelation In Cancer", by Arthur Furst. Also available from amazon: Chemistry Of Chelation In Cancer.
Carcinogenic Compounds And Chelating Agents
A large variety of chemicals are implicated as causative agents for cancer. At the present state of knowledge there is no evidence that the original agent applied is itself the primary carcinogen; a metabolic product may be the true carcinogenic agent. Many workers are now investigating the metabolic pathway of the agents which produce cancer when applied topically or given parenterally to experimental animals. What is accepted by many is that normal cells, when exposed to a carcinogen, go through a precancerous state and then become malignant. All carcinogens appear to bind both soluble proteins and nucleic acids. This binding may be more complicated than simple solubility of the agent in tissues or dermal lipids for protein or nucleic acid complexes may be involved, and so may electron transfer processes. Metals can mediate electron transfer mechanisms. These in vivo bindings of carcinogens by proteins may lead to a deletion of proteins necessary for enzymatic control of growth (379, 562).
Carcinogenesis is a complex process and may be multistage. The two stage hypothesis has much to offer. For rapid formation of malignancies there can be an initiator, like urethane, and a promotor, like croton oil.
In the usual classification of substances which have any effect on cancer, a distinction is usually made between carcinogens and anti cancer compounds. Little notice is ordinarily taken of "Had dow's paradox" that an anti cancer compound may also be a carcinogen. Thus, carcinogenic hydrocarbons have some beneficial effect in breast cancer, whereas triethylenemelamine is carcinogenic.
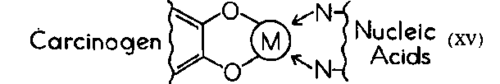
The one property which most of these agents have in common, be they carcinogenic or anti tumor, is their ability to bind metals, or to bind to tissues via metals (XV). The chelation hypothesis takes cognizance of the fact that carcinogens or antitumor agents, despite dissimilar biological results, may act by similar mechanisms.
Continue to:


