Alcohols and Acid Lipoids
Description
This section is from the book "Research In Physiopathology As Basis Of Guided Chemotherapy With Special Application To Cancer", by Emanuel Revici. Also available from amazon: Research In Physiopathology
Alcohols and Acid Lipoids
The role of the relationship between polar and nonpolar groups appeared very clear in the study of the series of aliphatic alcohols.
We have considered separately a group of agents extensively used in our research. These are organic substances that have a radical with a bivalent sulfur as the polar group. We have mentioned above that an inorganic substance with similar constitution, sodium thiosulfate, has a strong alkalizing effect upon the s.d.c. pH. Such an effect upon the s.d.c.
pH has been obtained with all preparations containing a polar thiol group when administered orally or parenterally. We must emphasize the unusual uniformity of results, rarely encountered in other biological experiments. It appears that the alkalizing effect of these preparations is sufficient to override the individual differences in subjects receiving them. The same alkalizing effect has been apparent for sodium thiosulfate, and seems to be a common characteristic for substances having a bivalent sulfur in their polar group.
These alcohols exert no influence upon the pH of normal tissues, as seen in measurements taken immediately after the skin is cut. The first members of the series, the methyl to propyl alcohols, do not influence the s.d.c. pH. From butyl alcohol to nonyl alcohol, the members of the series show a consistent acidifying influence. However, a very important observation was made when the four isomers of butyl alcohol were studied. Three showed the acidifying effect while one, the tertiary isomer, like the lower alcohols, did not influence the second day wound crust pH. This could be correlated to a special characteristic of these substances, their relative solubility in water and in neutral solvents. Just as do the lower alcohols, tertiary butyl alcohol mixes with water and neutral solvents, while the other three butyl alcohols, like the higher members of the series, are more soluble in neutral solvents than in water. (Fig. 236) This characteristic, which was used in systematizing the polar nonpolar substances, appears to determine the activity of the aliphatic alcohols upon the s.d.c. pH. We must again emphasize that this activity is not direct but influences certain metabolic processes, since the active alcohol induces local acidification rather than the alkalization to be expected with a direct effect.
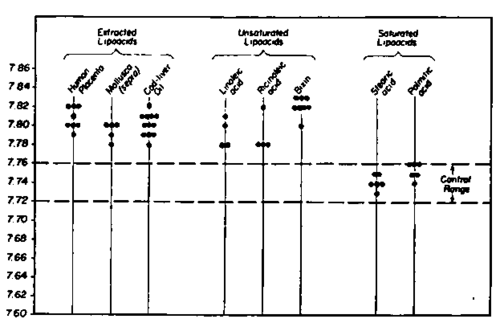
Fig. 235. The administration of various lipoacids upon the s.d.c. pH shows that while the saturated fatty acids do not influence it, the non saturated fatty acids as well as the lipoacid preparations obtained from different sources induce an elevation of the local pH.
Another factor which seems to influence the activity of this alcohol series is the length of the carbon chain. While heptanol induces the characteristic acidification, octyl alcohol does so in only some animals. Nonyl and decyl alcohols appear inactive.
The possibility of a biological competition between these agents and fatty acids has led to research with other alcohols capable of combining with fatty acids, particularly in vivo. We studied glycerol, glycerophos phoric ion and sterols, which are frequently found combined with fatty acids. We added glucose to this group because of its metabolic relationship to glycerol derivatives, although it is apparently not related to fatty acids.
It is interesting to note that glycerol produced only minimal influence upon the immediate pH. The highest values were still in the normal range. The acidifying effect of glycerol upon the s.d.c. pH, seemed to be subject to individual variations but also with values at the lower limit of normal, such as 7.72. Glucose showed a slight acidifying effect, while cholesterol showed definite acidifying activity upon the s.d.c. pH, as did all the preparations of the insaponifiable fraction of various organs or tissues. The acidification produced by the insaponifiable fraction preparations is more intense than for cholesterol alone, indicating that the other constituents of these preparations also have an acidifying influence.

Fig. 236. Second day wound crust pH values for various alcohols shows the relationship between activity and lipoidic property. All the alcohols below butanol and tert. butanol, which are not lipoids, are inactive. This also holds true for alcohols with chains longer than eight carbons.
Acid Lipoids
The next step in the use of the s.d.c. pH was the study of the effects of a special group of acids in which we were interested, the fatty acids. Analysis of the results obtained clearly shows the importance of the nonpolar group. A carboxyl, when bound to a long chain as in the fatty acids, does not by itself seem able to induce a change in the second day wound crust pH, the values remaining in the normal range. The substance appears inactive when the nonpolar group does not have its own energetic center or formation. The saturated fatty acids with 10-16 carbons do not influence the s.d.c. pH. The presence of double bonds in the nonpolar group changes the influence exerted. All the nonsaturated fatty acids studied show an alkalizing effect, with relatively slight differences for the higher desaturated members such as linolenic or arachidonic fatty acids, or for the fatty acid mixture obtained from cod liver oil. However, the conjugated fatty acids studied, such as eleostearic acid, or the mixture of acids starting from cod liver oil, showed the highest values in this group, even for so small an amount as 5 mgr. per animal per day. These data indicate the role of energetic formations in the nonpolar groups of fatty acids.
We must emphasize the difference between the hydrosoluble organic acids and the group of fatty acids mentioned above. In the former, the action seems due to the intervention of the carboxyl, probably explaining the strong activity of tricarboxylic citric acid. With an important nonpolar group, this carboxyl seems to be unable to carry the molecule, and therefore cannot act. The intervention of a double bond could serve in two ways: 1) by bending the molecule, thus increasing its mobility as it also reduces the melting point; 2) as an energetic center where reactions take place. The fact that the 10 carbon lauric acid is inactive would indicate the slight influence to be expected from the bending of the molecule alone, as in oleic acid. The influence exerted by metabolic changes in which fatty acids intervene through the energetic centers in the nonpolar group explains the fact that they have a local alkalizing effect instead of the acidifying effect of many other organic acids. (Fig. 235)
Continue to:


