Conjugated Fatty Acids And Quenching
Description
This section is from the book "Research In Physiopathology As Basis Of Guided Chemotherapy With Special Application To Cancer", by Emanuel Revici. Also available from amazon: Research In Physiopathology
Conjugated Fatty Acids And Quenching
Nonconjugated fatty acids such as linoleic acid, linolenic acid, arachidonic acid, mixed fatty acids from body liver oil and cod liver oil have a limited quenching action. Conjugated dienes such as isomers of linoleic acid or conjugated trienes such as eleostearic acid obtained through conjugation of linolenic acid or extracted from China wood oil also have a limited quenching effect upon hydrocarbon carcinogens and related compounds. (Table XXXVII) The same is true for mixtures of conjugated dienes and trienes.
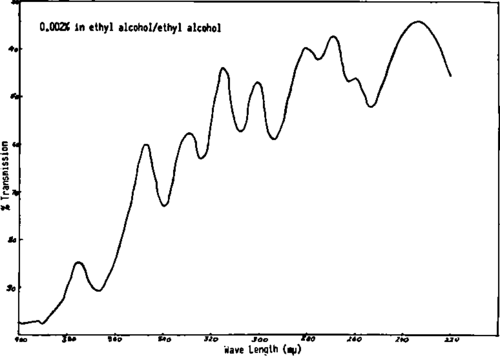
Fig. 281. Spectral analysis of conjugated cod liver oil fatty acids shows the presence of conjugated di-, tri-, tetra-, penta- and hexaenes.
Fatty acid mixtures having conjugated di-, tri-, tetra-, penta- and hexaenes as shown by spectral analysis (Fig. 281) have been found to exhibit a high degree of quenching activity (Fig. 282) (Table XXXVII) when mixed with hydrocarbon carcinogens.
The quenching action of fatty acids upon the fluorescence of hydrocarbon carcinogens appears to be nonadditive. When the incident ray is passed first through an 0.2% solution of conjugated fish oil fatty acids in alcohol and then through an 0.012% solution of methylcholanthrene in alcohol in separate vessels, the residual fluorescence is 81%. When the same two solutions are mixed together in one cell, the residual fluorescence is 11.2%.
The relationship of the different conjugated members to the quenching effect has been studied. Fatty acid mixtures having different proportions of isomers with 2, 3, 4, 5 and 6 conjugated double bonds were obtained by conjugation or by treatment of conjugated mixtures with heat, oxygen, chlorine or sulfur. Changes in the proportions of the di-, tri-, tetra-, penta and hexaenes were followed by means of spectral analyses. The changes in the height of the peaks in these curves corresponding to the different conjugated polyenes were then compared with the changes in the quenching effect of the corresponding fatty acid mixtures. Figure 283 shows the spectral analysis of samples obtained at various intervals during the action of oxygen upon a mixture of conjugated fatty acids. As seen, oxygen induces unequal changes in the height of the peaks in the curves of which correspond to di-, tri-, tetra-, penta- and hexaenes. Fig. 284 shows the quenching activity of the mixtures. It can be seen that a parallelism exists between the relative proportions of the tetraenic component and the quenching activity of the mixtures. In this experiment it appears that the quenching effect could also be related to the presence of conjugated pentaenes. Evidence available from other experiments do not, however, sufficiently support this.

Fig. 282. Quenching of fluorescence of a methylcholanthrene (.0062%) solution in alcohol by different concentrations of conjugated fish oil fatty acids.
We studied in a similar way the effect induced by the treatmentwith sulfuric acidof a mixture of conjugated fatty acids of cod liver oil. Fig. 285 shows part of the occurring changes and Fig. 286, the quenching effect.
Similarly, we studied the changes in the quenching effect during the conjugation with KOH of cod liver oil fatty acids in ethyl alcohol. Fig. 287 shows the conjugation effect and 288, the quenching effect of the preparation at different moments, in various dilutions.
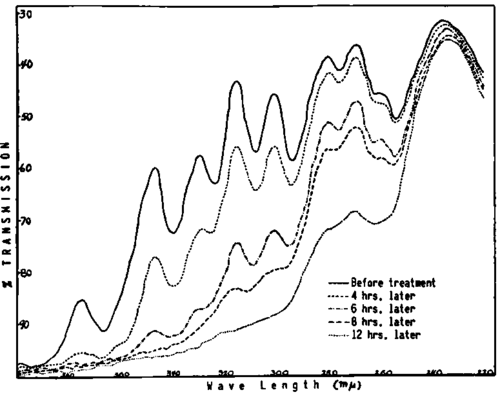
Fig. 283. Changes in the absorption spectra of a mixture of conjugated fish oil fatty acids induced by treatment with oxygen. The treatment has a greater effect on the higher unsaturated members, with the proportion of tri-, tetra-, penta-, and hexaenes decreasing as treatment continues, as seen by the reduction in the height or even disappearance of the peaks. After 12 hours of treatment, the conjugated dienes are the only ones not yet influenced. Dilution 0.002% in ethyl alcohol.
The entire problem was simplified by studying a pure conjugated tetraene. We have obtained pure tetraenic parinaric acid from akariton fat of Parinarium laurinum seeds. In addition, we have prepared almost pure tetraenes utilizing the technique described by Maury, Brode and Brown. Unfortunately, with the last method, the results were less favorable, the proportion of tetraenes beginning to decrease long before the conjugated dienes and trienes have disappeared. Pure tetraenic conjugated acid has shown that the quenching action is related almost entirely to the tetraenic component alone, and in a mixture it is largely parallel to the content in conjugated tetraenic fatty acids. Fig. 289 shows the quenching curve induced by parinaric acid.

Fig. 284. The relationship between changes of the di-, tri-, tetra-, penta- and hexaenic peaks as found on spectral analysis of samples of conjugated fish oil fatty acids treated for different lengths of time with oxygen and the quenching effect of the same samples. A close parallelism exists between the decrease in the proportion of tetra- and pentaenic peaks and the quenching activity of the mixture.

Fig. 285. The changes in the spectral analysis of a mixture of conjugated cod liver oil fatty acids, induced by the treatment with sulfuric acid. The treatment leads to unequal decrease in the amount of different conjugated members. Only two curves are shown: at the beginning of the treatment and at 260 minutes.
Continue to:


