Fluorinated Steroids
Description
This section is from the book "The Scientific Contributions Of The Ben May Laboratory For Cancer Research", by The University of Chicago. Also available from Amazon: The Scientific Contributions Of The Ben May Laboratory For Cancer Research.
Fluorinated Steroids
Since fluorine is a highly electronegative atom, it was hoped that steroid hormones in which a hydroxyl group is replaced by a fluorine atom would be rather similar in electronic and dipole characteristics to the original hormone and yet be incapable of participating in oxidation-reduction or hydrogen bonding reactions at that position. Furthermore, substitution of an electronegative fluorine atom for a hydrogen atom adjacent to a hydroxyl or ketone group might influence the contribution of that group to primary hormonal activity in a somewhat different way than it affects the contribution to secondary activities. In the hope that such modifications might furnish compounds in which primary hormonal potency was decreased without significant reduction of secondary physiological activities, considerable effort has been devoted to the development of methods for the introduction of fluorine into the steroid molecule. Novel fluorinated steroids prepared by these methods are listed in Table 5.
Table 5. Novel Fluorinated Steroids
2α-Fluorocholestan-3-one | 4-Fluorotestosterone |
2α-Fluoroandrostane-3,17-dione | 4β-Fluorocoprostan-3-one |
2α-Fluoro-17β-hydroxyandrostan-3- | 4β-Fluoropregnane-3,20-dione |
one | 4,4-Difluorocoprostan-3-one |
2,2-D ifluorocholestan-3-one | 4,4-Difluoro-5-cholesten-3-one |
2,2-Difluoro-17β-hydroxyandrostan- | 4,4-Difluoro-5-pregnene-3,20-dione |
3-one | 4,4-Difluoro-17β-hydroxy-5-andro- |
2,2,4o-Trifluoro-17β-hydroxyandro- | sten-3-one |
stan-3-one | 4,4-Difluoro-17β-hydroxy-l 7-methyl- |
3β-Fluoro-5-cholestene | 5-androsten-3-one |
3β-Fluoro-5-pregnen-2O-one | 6/3-Fluoro-4-cholesten-3-one |
3β-Fluoro-5-androsten-17β-ol | 17-Fluoroprogesterone |
3βFluoro-5-androsten-l 7-one | 17,21 -D ifluoroprogesterone |
3β-Fluoro-17-methyl-5-androsten- | 17,21,21-Trifluroprogesterone |
17β-ol | 21 -Fluoroprogesterone |
3β-Fluoroandrostan-17β-ol | 21,21-Difluoroprogesterone |
3β-Fluoroandrostan-17-one | 21-Fluoro-11β-hydroxyprogesterone |
3β-Fluoro-l 7-methylandrostan-17β-ol | 21 -Fluoro-21 -deoxy cortisone |
4-Fluoro-4-cholesten-3-one | 21 -Fluoro-21 -deoxy Cortisol |
During the early stages of these investigations it was reported (Fried, J., and Sabo, E. F. J. Am. Chem. Soc, 76:1455, 1954) that a 9α-fluoro sub-stituent adjacent to the 11-oxygen function in the adrenal cortical hormones markedly increases biological activity. Although potentiation of primary hormonal activity was just the opposite of our intended objective in the sex hormone series, this finding substantiated the concept that a fluorine substituent could strongly influence the biological properties of a steroid.
Although simple cyclic olefins, such as cyclopentene and cyclohexene, readily add the elements of hydrogen fluoride, attempts to prepare fluoro-steroids by the addition of hydrogen fluoride to steroidal double bonds proved unsuccessful. A general method for the replacement of hydroxyl groups by fluorine was developed; this worked well for simple alcohols but failed with hydroxysteroids. This procedure involves reaction of the alcohol with phosgene to give the chloroformate ester and treatment of the latter substance with thallous fluoride to give the fluoroformate ester, which, on heating with either pyridine (146) or boron trifluoride (147), eliminates carbon dioxide to produce the alkyl fluoride in good yield. Although the fluoroformate esters of a number of 3- and 17-hydroxysteroids were prepared without difficulty, these did not undergo conversion to fluorosteroids under the conditions investigated.
Treatment of the i-methyl ether of cholesterol with hydrogen fluoride in acetic acid solution afforded cholesteryl fluoride, the first fluorinated steroid to be synthesized. However, the yield was so low as to render this method impractical. A satisfactory procedure was developed for the preparation of 3β fluorosteroids, which involves conversion of a 3β-hydroxy-Δ5-steroid to the corresponding 3-iodocompound (by treatment of its p-toluenesulfonate ester with magnesium iodide) followed by reaction of the iodosteroid with anhydrous silver fluoride in acetonitrile-xylene solution (109). Catalytic hydrogenation of the 5,6-double bond gives saturated 3 β-fluorosteroids. 21-Fluorosteroids were obtained by converting the 21-hydroxyl group of a 20-ketopregnane compound to the corresponding 21-iodosteroid and treatment of the latter substance with aqueous silver fluoride in acetonitrile solution (185).
Replacement by fluorine of hydrogen atoms adjacent to ring carbonyl group was accomplished by novel reactions of a recendy available reagent, perchloryl fluoride. Certain properties of this substance suggested to us that, in the absence of acids, perchloryl fluoride can react as if fluorine is electrophilic, representing the first example of this type of fluorine activity. Accordingly, the reaction of perchloryl fluoride with such electron-rich groupings as enamines and enol ethers was investigated and found to furnish an extremely useful procedure for the fluorination of steroids.
Enamines (20) and enol ethers (145) derived from saturated 3-ketoster-oids with the 5 a-configuration react rapidly at low temperature with perchloryl fluoride in pyridine or pyridine-ether solution to yield 2α-fluoro-3-ketosteroids (Fig. 28, eq. 1). The process, preferably using the enamine, may be repeated, resulting in a stepwise replacement of the hydrogen atoms adjacent to the carbonyl group by fluorine. Enol ethers and enamines of 3-ketosteroids of the 5β-series react similarly with perchloryl fluoride to yield ketosteroids fluorinated in position 4.
In the case of 3-keto-Δ4-steroids, the reaction of perchloryl fluoride with the enamine takes a different course from that with the enol ether. The enamine yields a mixture of 4-fluoro-Δ4- and 4,4-difluoro-Δ5-steroids (eq. 3), with the proportion of each depending on the reaction conditions (144)- Enol ethers of 3-keto-Δ4-steroids are fluorinated in position 6 (eq. 2). This reaction (145) affords an improved synthetic route to the previously known 6-fluorinated steroids in the progestational and cortical hormone series, some of which have greatly enhanced biological activity.

Fig. 28.-Reaction of perchloryl fluoride with steroid enamines and enol ethers.
In contrast to enol ethers, the enol esters of steroid ketones are relatively unreactive toward perchloryl fluoride. But with certain steroid ketones, for which it was not possible to prepare an enamine or enol ether derivative, the reaction of the enol acetate with perchloryl fluoride did furnish the fluorinated steroid in rather low yield. 17-Fluoroprogesterone, 17,21-difluoroprogesterone, and 16β-fluoro-4-androstene-3,17-dione were obtained in this way.
Stilbestrol And Kexestrol Analogues
As far as is known, the synthetic estrogens, stilbestrol and hexestrol (Fig. 29, IIand III; Ar = Ar' =p-HOC6H4,-), exhibit all the physiological actions of the natural steroid estrogens. To investigate the effect of alterations of the aromatic rings on hormonal activity as compared to that on secondary physiological actions, a general synthetic procedure was developed by which the chemical nature of one or both rings can be varied as desired. This process involves the conversion of an aromatic aldehyde to the corresponding 4-arylhexan-3-one (142), which on treatment with an aryllithium reagent yields the carbinol, I. Acid-catalyzed dehydration of I gives the stilbestrol analogue, II(140), whereas catalytic hydrogenolysis of I takes place with retention of configuration to furnish the hexestrol analogue, III (151)- Using this procedure, the substances listed in Table 6 were prepared and subjected to biological evaluation.
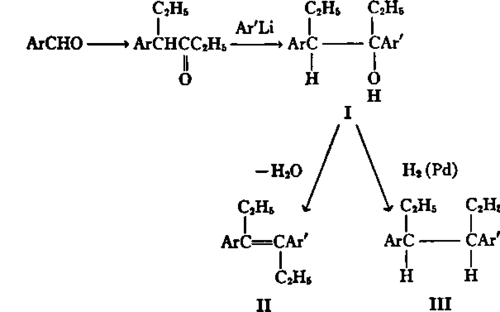
Fig. 29.-Synthesis of stilbestrol and hexestrol analogues.
Continue to:


