Mechanisms Of Hormone Action. Part 8
Description
This section is from the book "The Scientific Contributions Of The Ben May Laboratory For Cancer Research", by The University of Chicago. Also available from Amazon: The Scientific Contributions Of The Ben May Laboratory For Cancer Research.
Mechanisms Of Hormone Action. Part 8
DPNH | Pyridine aldehyde-DPN | Rate of redu acceptor nu (µmoles/m protei ction of cleotide in./mg.n) | |
(umoles) | (umoles) | No steroid | With steroid |
0.4 | 1.25 | 1.45 | 6.16 |
0.4 0.4 0.4 | 2.50 3.75 5.00 | 1.96 1.74 1.43 | 9.64 12.3 10.9 |
* Reactions were carried out at 25 C. in final volumes of 3.0 ml. containing: 100 µmoles Tris of pH 8.5; 20 µg. 4-androstene-3,17-dione in 0.03 ml. methanol or 0.03 ml. methanol only; stated quantities of DPNH and pyridine aldehyde-DPN; and 94 (ig. of β-hydroxysteroid dehydrogenase (specific activity 11,200 units per milligram protein). Measurements were made at 385 mµ, and the molar extinction coefficient was assumed to be 4130.
2. Liver 3a.-Hydroxy Steroid Dehydrogenase
Rat liver contains two pyridine nucleotide-linked 3a-hydroxysteroid dehydrogenases localized in the soluble cell sap and in the microsomal particles, respectively (55, 56, 120). The particulate and soluble enzymes both react at nearly equal rates with DPN and TPN. Soluble 3a-hydroxysteroid dehydrogenase has been partially purified and some of its properties examined by Tomkins (120). We have found that this enzyme reduces the acetyl-pyridine analog of DPN, as well as DPN and TPN, in the presence of substrate amounts of 3a-hydroxysteroids. Slightly purified preparations of soluble rat liver 3a-hydroxysteroid dehydrogenase have been shown to catalyze a steroid-dependent hydrogen transfer from TPNH to DPN and from DPNH to the acetylpyridine analog of DPN (55). Low concentrations (10-6M or less) of 3a-hydroxy- and 3-ketosteroids, which are substrates for this enzyme, are capable of promoting this transhydrogenation.
Efficient transhydrogenation by this enzyme depends upon a critical balance of donor and acceptor nucleotides. Very small concentrations of TPN, for which the enzyme has high affinity, may entirely suppress the hydrogen transfer from DPNH to the acetylpyridine analog of DPN.
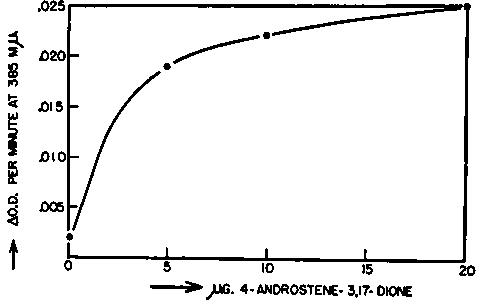
Fig. 6. Velocity of hydrogen transfer between DPNH and the pyridine aldehyde analog of DPN catalyzed by purified bacterial B-hydroxysteroid dehydrogenase in the presence of varying concentrations of 4-androstene-3,17-dione.
The measurements were carried out at 25° C. in cuvettes of 1.0 cm. light path. The reaction systems contained in a final volume of 3.0 ml.: 100 umoles Tris buffer of pH 8.8, 0.45 umole DPNH, 4.6 umoles of the pyridine aldehyde analog of DPN, varying amounts of 4-androstene-3,17-dione in 0.03 ml. methanol, and 94 ug. (900 units) of purified bacterial B-hydroxysteroid dehydrogenase. The rates were derived from initial optical density changes with time at 385 mu.
Furthermore, the transfer of hydrogen from TPNH to DPN is demonstrable only when the total concentration of [TPN + TPNH] is low compared to the concentration of [DPN + DPNH].
The demonstration of steroid-dependent hydrogen transport in soluble liver systems required a certain degree of purification of the 3a-hydroxy-steroid dehydrogenase to minimize the interference by other enzymes and to reduce the quantities of bound pyridine nucleotides.
Much evidence supports the view that the soluble 3a-hydroxysteroid dehydrogenase of fiver shuttles hydrogen by virtue of an alternate oxido-reduction of the steroid, and that in this instance also, the steroid hormone acts in the manner of a hydrogen carrier or coenzyme. With catalytic quantities of androsterone or androstane-3,17-dione, this process may be represented thus:
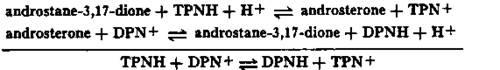
V. Distribution And Functional Significance Of Pyridine Nucleotide Transhydrogenases
The existence of enzymatic pathways for hydrogen transport between DPN and TPN was foreshadowed by Ball in 1941 (5), who suggested that flavoproteins might carry out this function. But it was not until 1952 that Colowick, Kaplan, and their collaborators provided evidence for distinct pyridine nucleotide transhydrogenases.
1. Pseudomonas Fiuorescens Transhydrogenase
During a study of the isocitric dehydrogenase activity of soluble extracts of Pseudotnonas fiuorescens, Colowick et al. (20) observed that DPN was not reduced by this TPN-specific enzyme. However, a rapid reduction of DPN by isocitrate occurred if catalytic quantities of TPN were added to the reaction mixture. This led to the discovery of a new enzyme, designated pyridine nucleotide transhydrogenase, which catalyzes, inter alia, the following reaction:
TPNH + DPN+ ⇋ TPN+ + DPNH
Kaplan, Colowick and their co-workers purified this transhydrogenase more than 200-fold (20), and showed conclusively that it catalyzed a reaction between intact pyridine nucleotide molecules in which a direct and stereospecific transfer of hydrogen took place (63, 102). The possibility of an exchange of phosphate, or of other moieties (e.g., of the pyridinium ring) was eliminated (66). The purest preparations of the enzyme catalyzed the transfer of 11-12 umoles of hydrogen per milligram protein per minute at 25° C.
Kaplan et al. also showed that deamino-DPN and deamino-TPN could serve either donor or acceptor functions with respect to either DPN or TPN and that nicotinamide mononucleotide and ribosyl nicotinamide would function as acceptors (63, 65). With nicotinamide C14-labeled DPN (66), they found that the enzyme catalyzed an exchange reaction between DPNH and DPN. They considered the possibility that the purified Pseudotnonas transhydrogenase was a dehydrogenase whose substrate(s) reacted with these forms of pyridine nucleotide and effected hydrogen transfer by a coupled oxidoreduction with catalytic amounts of substrate. Three reasons rendered this unlikely: (a) purified transhydrogenase preparations did not reduce either DPN or TPN in the presence of the following substrates: glutamate, isocitrate, lactate, glucose-6-phosphate, 6-phosphoglu-conate, and ethanol; (b) the purified enzyme was so active that it seemed improbable that the small quantities of enzyme added to the reaction mixture would contain adequate amounts of bound substrates; (c) transhydrogenation between DPN and TPN could not be demonstrated with two purified enzymes which react with both of these nucleotides, namely, lactic and glutamic dehydrogenases. Furthermore, neither alcohol nor lactic dehydrogenase catalyzed hydrogen transfer between DPN and deamino-DPN. However, the DPNH-DPN exchange reaction was catalyzed by alcohol dehydrogenase in the presence of ethanol (66).
2. Particulate Transhydrogenases Of Animal Tissues
The pyridine nucleotide transhydrogenase activity of mammalian mitochondria was described by Kaplan, Colowick, and Neufeld (64), who isolated a partially purified enzyme from beef heart particles by treatment with digitonin. The mitochondrial transhydrogenase of heart differs from the Pseudomonas enzyme in many important respects. Thus, the oxidation of TPNH by DPN catalyzed by the mitochondrial enzyme is freely reversible, whereas the reverse reaction barely proceeds in the presence of the bacterial transhydrogenase unless 2'-AMP is added (65). There are certain differences in specificity. The bacterial enzyme promotes the oxidation of TPNH by deamino-TPN, whereas the beef heart transhydrogenase does not catalyze this reaction. The latter system, unlike the Pseudomonas enzyme, is not influenced by inorganic phosphate. Evidence for the transhydrogenase activity of the mitochondria of many animal tissues is now available (54, 67, 92, 124).
The mitochondria also catalyze an exchange reaction between DPNH and DPN, like the Pseudomonas transhydrogenase, but it has been recently suggested by Stein and colleagues (105) that the latter reaction in animal tissues is catalyzed by a different enzyme from the TPNH-DPN transhydrogenase. The DPNH-DPN exchange system is also found in liver microsomes, which appear not to catalyze the TPNH-DPN transhydrogenase reaction. In earlier experiments, Weber and Kaplan (133) showed that a number of flavoproteins will catalyze the transfer of hydrogen from TPNH and DPNH to pyridine nucleotide analogs of a higher oxidation-reduction potential (e.g., the acetylpyridine analog of DPN), but not between the two natural forms of pyridine nucleotide.
Continue to:


