Protocols. Prostatic Cancer
Description
This section is from the book "The Scientific Contributions Of The Ben May Laboratory For Cancer Research", by The University of Chicago. Also available from Amazon: The Scientific Contributions Of The Ben May Laboratory For Cancer Research.
Protocols. Prostatic Cancer
Case 1.-E.S., age 63, atrophy of primary prostatic cancer with active osseous metastases. Orchiectomy May, 1947, with recurrence of pain in back and rectum in 1950. Complete relief of pain after adrenalectomy in February, 1951. Urinary retention October, 1951, treated by total perineal prostatectomy; no cytologic evidence of active cancer cells in excised prostate. Gained 8.4 kg. in 251 days.
Case £.-C.F., age 47, extensive neoplastic involvement of prostate, bones, and lymph nodes. Orchiectomy, 1948. On admission, March, 1951, moribund with massive edema of both legs and external genitalia; large constricting peri-rectal and abdominal masses; uncontrollable bone pain; bed-ridden and emaciated. After adrenalectomy cessation of pain; shrinkage of neoplastic masses. Gained 16 kg. in 230 days.
Case 3.-J.S., age 59. Died 72 hours after operation.
Case 4--L.S., age 62, active primary tumor and metastases.
Orchiectomy, 1946. For 2 years severe pain in legs requiring narcotics; urinary frequency and retention; large indurated neoplastic mass in seminal vesicle; extensive osseous metastasis. Following adrenalectomy no decrease in urinary retention; slight decrease in size of neoplastic mass; complete relief of pain. Died of pulmonary edema 49 days after adrenalectomy.
Case 5.-W.M., age 62, active primary prostatic neoplasm and osseous metastasis. Severe pain in back persisted after orchiectomy 60 days before. Prostate enlarged and indurated. Following adrenalectomy disappearance of pain, complete regression of prostatic enlargement as determined by palpation. Gained IS kg. in 180 days.
Case 6.-J.F., age 59, active primary prostatic cancer with active osseous metastases. Orchiectomy April S, 1950. In December, 1950, recurrence of severe sciatica poorly controlled by narcotics; osseous metastasis; prostate enlarged ++ and indurated; bed-ridden. Following adrenalectomy there was complete relief of pain and regression of the prostate to atrophic condition as determined by palpation. Returned to work. Gained 4 kg. in 158 days.
Case 7.-G.H., age 53, active primary prostatic cancer and osseous metastasis. Severe pain in shoulders, ribs, pelvis, and legs. Walnut-sized, hard nodule in prostate. No significant modification of neoplastic disease by adrenalectomy. Gained 4 kg. in 137 days.
Mammary Cancer
Case 8.-L.C., female, age 41. Died 48 hours after adrenalectomy.
Case 9.-M.B., female, age 47, extensive osteolytic metastases to pelvis and lymphedema of arm. Radical mastectomy in 1949; severe pain in pelvis and leg with osseous metastasis, November, 1950; rapid progression of the osteolytic lesion despite treatment with testosterone propionate and irradiation of pelvis. Following adrenalectomy partial relief of pain but continued advance of osseous lesions. Ovariectomy 56 days after adrenalectomy. Slight sclerosis of osteolytic lesions and considerable but not complete relief of pain. Gained 5 kg. in 168 days.
Case 10.-R.B., female, age 57, pulmonary metastases with dyspnea. No clinical improvement after adrenalectomy. Died 23 days after adrenalectomy.
Case 11.-J.B., female, age 40, large ulcerating mammary lesions with lymphedema of arm. Adrenalectomy followed by rapid advance of ulcerations.
Case 12.-G.N., female, age 43, hemothorax and metastasis to spine and pelvis. Radical mastectomy, 1945. Pain in spine with collapse of Tu vertebra and extensive destructive lesions in pelvis in June, 1950. Treated with testosterone propionate with complete relief of symptoms until May, 1951, when massive accumulation of fluid in pleural cavity occurred requiring repeated thoracentesis. Right adrenalectomy and bilateral ovariectomy, 6/13/51; left adrenalectomy, 6/28/51. No recurrence of pleural fluid of magnitude sufficient to require tapping in 155 days (Figs. 7,8). Complete relief of symptoms.
Case 13.-N.C., male, age 53, intracranial and pulmonary metastasis. Radical mastectomy. May, 1948. In June, 1950, metastases found in routine chest roentgenogram. Bilateral orchiectomy, 11/25/50, followed by further progression of pulmonary lesions. For 3 months, anorexia and vomiting; for 3 weeks, diplopia. Adrenalectomy was followed by disappearance of neurologic signs and double vision. Regression of pulmonary lesions on x-ray examination 39 days (Figs. 1, 2) after operation. Gained 10 kg. in 117 days.
Case 14--L.M., female, age 65. Carcinoma of right breast with cutaneous metastases in scar of left radical mastectomy (12/2/48). A large indurated mass was found in the right breast with extensive, red infiltrations in mastectomy scar. Within 4 weeks titer adrenalectomy flattening and pallor of cutaneous lesion (Figs. 3, 4) and decrease in size of unoperated tumor in right breast.

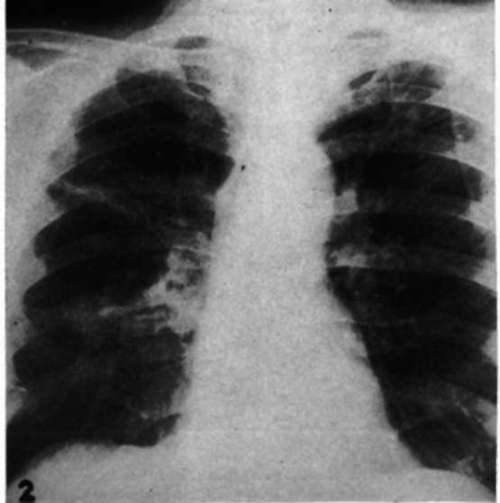
Fig. 1.-Metastases to both lungs from mammary cancer in a male (Case 13) before adrenalectomy.
Fig. 2.-Roentgenogram of chest of a male with mammary cancer (Case 13) 39 days after adrenalectomy.

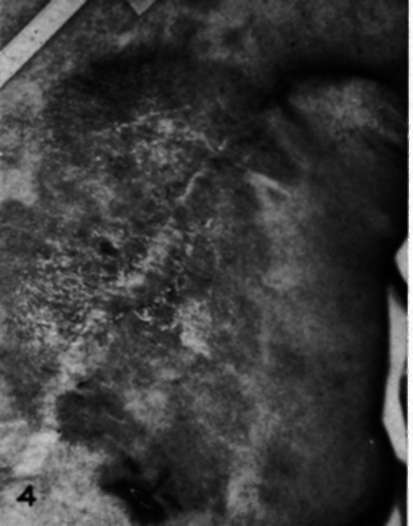

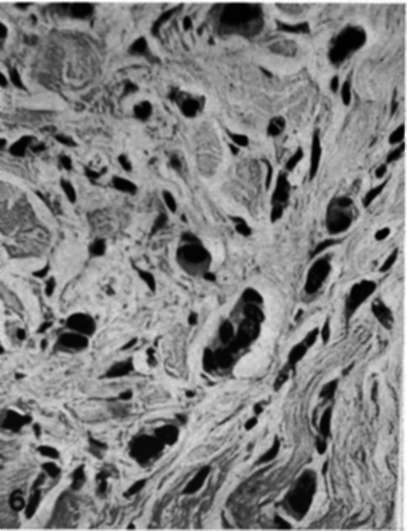
Fig. 3.-Recurrent carcinoma in a mastectomy scar in a woman (Case 14) before adrenalectomy. Fig. 4.-Appearance of mastectomy scar (Case 14) 40 days after adrenalectomy.
Fig. 5.-Biopsy from dermal plaque of recurrent mammary cancer in a mastectomy scar (cf. Fig. 3) showing adenocarcinoma. (Dr. E. M. Humphreys.) X475.
Fig. 6.-Biopsy from dermal plaque of recurrent mammary cancer in a mastectomy scar (cf. Fig. 4) 35 days after adrenalectomy showing an abundance of cancer cells which are shrunken with darkly stained nuclei. (Dr. E. M. Humphreys.) X475.
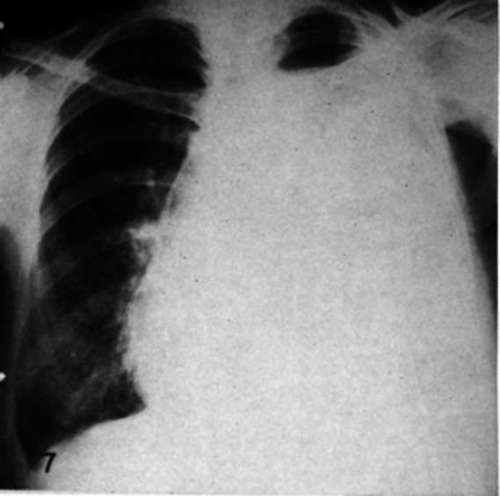
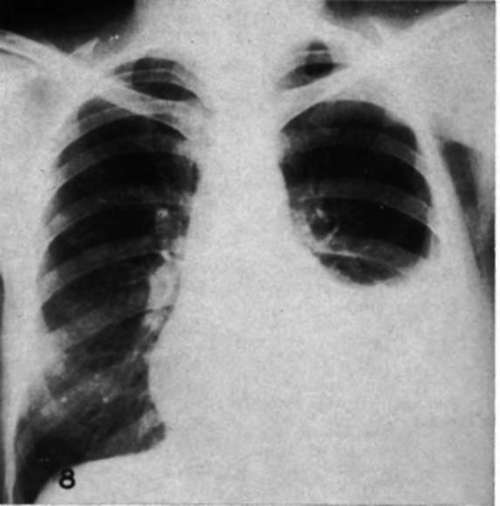
Fig. 7.-Metastatic carcinoma of the breast causing hemothorax in a woman (Case 12) before adrenalectomy.
Fig. 8.-Roentgenogram of thechestof a woman with metastatic mammary cancer (Case 12) 116 days after adrenalectomy. Thoracentesis has not been done since adrenalectomy.
Miscellaneous Cancers
Cass 16.-A.K., male, age 58. An extensive squamous carcinoma of the urethra with metastasis to lymph glands of both inguinal regions and the right tibia was not benefited by bilateral adrenalectomy. Died 03 days after operation.
Cats 16.-M.S., female, age 53. A very widely spread melanosarcoma with extensive pulmonary metastases was not improved after adrenalectomy. A great increase in sixe of pulmonary metastases was observed in roentgenograms made 4 weeks after operation.
Cass 17.-J.S., male, age 41. Very large bilateral pulmonary metastases from a chorionic epithelioma of the testis was not affected favorably by bilateral adrenalectomy within 4 weeks after adrenalectomy.
Cass 18.-M.N., female, age 22. An extensive undifferentiated carcinoma involving the right lung and left cervical lymph nodes continued to grow rapidly 4 weeks after adrenalectomy.
References
1. Adair, F. E., and Herrman, J. B. The Use of Testosterone Propionate in the Treatment of Advanced Carcinoma of the Breast. Ann. Surg., 123:1023-35, 1046.
2. Bischoff, F., and Maxwell, L. C. Hormones in Cancer. V. The Effect of Glandular Extirpation upon the Growth of Transplantable Tumors. J. Pharm. 4c Exper. The rap., 46:51-57, 1932.
3. Brzcstowicz, R. J.; Svten, H. J.; Bennett. W. A.; and Hiqgins, G. M. The Effect of Cortisone on the Growth of Transplanted Ependymomas in Mice. Proc. Staff Meetings Mayo Clinic, 26:121-28,1951.
4. Cox, H. T. Adrenalectomy and Prostatic Cancer: Report of Three Cases. Lancet, 2:425-26, 1947.
5. Ebebsole, C. M., and Huggins, C. Effect of Cortisone Acetate on Prostatic Secretion in the Dog. Unpublished observations.
6. Eli el, L. P., and Pearson, O. H. The Clinical and Physiological Effects of Adrenocorticotrophic Hormone and Cortisone Acetate in Patients with Neoplastic Disease. New York State J. Med., 61:1839-43, 1951.
7. Emerson, G. A.; Wurtx, E.; and Zanetti, M. E. Regression of Lymphosarcoma Transplants Following the Administration of Cortisone to Riboflavin-deficient Mice. Fed. Proc., 9:357-58, 1950.
8. Farrow, J. H., and Adair, F. E. Effect of Orchidectomy on Skeletal Metastases from Cancer of the Male Breast. Science, 96:654, 1942.
9. Phaser, R. W.; Albright, F.; and Smith, P. H. The Value of the Glucose Tolerance Test, the Insulin Tolerance Test, and Glucose-Insulin Tolerance Test in the Diagnosis of Endocrinologic Disorders of Glucose Metabolism. J. Clin. Endocrinol., 1:297-306, 1941.
10. Funk, C; Tomashefskt, P.; Soukup, R.; and Ehrlich, A. The Effect of Hormonal Factors and the Removal of Certain Organs upon the Growth of a Transplanted Rat Tumour. Brit. J. Cancer, 6:280-87, 1951.
11. Haddow, A.; Watkinson, J. M-; Paterson, E.; and Kolleb, P. C. Influence of Synthetic Oestrogens upon Advanced Malignant Disease. Brit. M. J., 2:393-98, 1944.
12. Heilman, F. R., and Kendall, E. C. The Influence of 11-Dehydi^l7-hydroxycorticosterone (Compound E) on the Growth of a Malignant Tumor in the Mouse. Endocrinology, 34:416-20,1944.
13. Hiqgins, G. M.; Woods, K. A.; and Bennett, W. A. The Influence of Cortisone (Compound E) upon the Growth of a Transplanted Rhabdomyosarcoma in C3H Mice. Cancer Research, 10:203, 1950. 14. Huggins, C. Effect of Orchiectomy and Irradiation on Cancer of the Prostate. Ann. Surg., 116:1192-1200, 1942. 16.-. Prostatic Cancer Treated by Orchiectomy: The Five Year Results. J.A.M.A., 181:676-81, 1946.
16. Huggins, C, and Baker, R. Use of Serum Proteins in Measuring Activity of Prostatic Cancer. Cancer Research, 11:428-31, 1951.
17. Huggins, C, and Bergbnstal, D. M. Surgery of the Adrenals. J.A.M.A., 147:101-6, 1951.
18. Huggins, C; Cleveland, A. S.; and Jensen, E. V. Thermal Coagulation of Serum in Diagnosis. J.A.M.A., 143:11-15, 1950.
19. Huggins, C, and Hodges, C V. Studies on Prostatic Cancer. I. The Effect of Castration, of Estrogen and of Androgen Injection on Serum Phosphatases in Metastatic Carcinoma of the Prostate. Cancer Research, 1:293-97, 1941.
20. Huggins, C, and Scott, W. W. Bilateral Adrenalectomy in Prostatic Cancer: Clinical Features and Urinary Excretion of 17-Ketosteroids and Estrogen. Ann. Surg., 122: 1031-41, 1946.
21. Ingle, D. J., and Baker, B. L. The Effect of Adrenalectomy in the Rat upon the Rate of Growth of Transplantable Tumors. Endocrinology, 48:816-16,1951.
22. Ingle, D. J.; Prestbud, M. C; and Rice, K. L. The Effect of Cortisone Acetate upon the Growth of the Walker Rat Carcinoma and upon Urinary Non-Protein Nitrogen, Sodium, Chloride and Potassium. Endocrinology, 46:510-13, 1950.
23. Jo anno vies, D. G. fiber das Wachstum der transplan-tablen M&usetumoren in kastrierten und in epinephrek-tomierten Tieren. Beitr. path. Anat., 62:194-203, 1916.
24. King, E. J., and Armstrong, A. R. Convenient Methods for Determining Serum and Bile Phosphatase Activity. Canad. M. A. J., 31:376-81,1934.
25. Law, L. W.; Bunker, Jr., L. E.; and Norris, B. A. Effect of Gonadectomy and Adrenalectomy on the Appearance and Incidence of Spontaneous Lymphoid Leukemia in C58 Mice. J. Nat. Cancer Inst., 8:167-59, 1947.
26. Levy, M. S.; Power, M H., and Kepler, E. J. The Specificity of the "Water Test*' as a Diagnostic Procedure in Addison's Disease. J. Clin. Endocrinol., 6:607-82,1946.
27. Mubpht, J. B., and Sturm, E. The Adrenals and Susceptibility to Transplanted Leukemia of Rats. Science, 98:568, 1943.
28. Pearson, O.H ; Elikl, L. P.; Rawbon, R. W.; Dobbiner, K.; and Rhoads, C. P. ACTH- and Cortisone-Induced Regression of Lymphoid Tumors in Man. A Preliminary Report. Cancer, 2:943-45, 1949.
29. Postlethwait, R. W.; MosELET, V.; McKee, K. T.; Murdoch, J. II.. and McCobd, W. M. ACTH and Cortisone in Advanced Carcinoma of the Digestive Tract. Cancer, 4:984-87, 1961.
30. Roffo, A. H. 1/influence de la capsule surrenale sur le developpement des tumeurs ches Irs animaux prives de cette capsule et ches ceux traites avec des produits capsu-laires. Neoplasmes, 9:888-60, 1930.
81. Spies, T. D.; Stone, R. E.; Lopes, G. G.; Milanes, F.; Toe a, R. L.; and Reboredo, A. Response to Adrenocorticotropic Hormone and Cortisone in Persons with Carcinoma, Leukaemia and Lymphosarcoma. Lancet, 2: 241-44, 1950.
32. Sturm, E., and Mubpht, J. B. The Effect of Adrenalectomy on the Susceptibility of Rats to a Transplantable Leukemia. Cancer Research, 4:384-88,1944.
33. Suoiura, K.; Stock, C. C; Dobbiner, K-; and Rhoads, C. P. The Effect of Cortisone and Other Steroids on Experimental Tumors. Cancer Research, 10:244-45, 1950.
34. Taylor, S. G., HI; Ayer, J. P.; and Morris, R. S. Jr. Cortical Steroids in Treatment of Cancer. Observations on Effects of Pituitary Adrenocorticotropic Hormone (ACTH) and Cortisone in Far Advanced Cases. J.A.M.A., 144:1058-64, 1950. 35. Welkins, L.; Lewis, r. A.; Klein, R.; Gardner,L. I.; Crioler, J. F.; Rosemberg, E.; and Migeon, C. J. Treatment of Congenital Adrenal Hyperplasia with Cortisone. J. Clin. Endocrinol., 11:1-25, 1951.
Continue to:


