The Influence Of The Ingestion Of Carbohydrate. The Respiratory Metabolism. Part 2
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
The Influence Of The Ingestion Of Carbohydrate. The Respiratory Metabolism. Part 2
The calorimetric and respiratory experiments which established these interrelations are portrayed in the accompanying chart (Fig. 20).
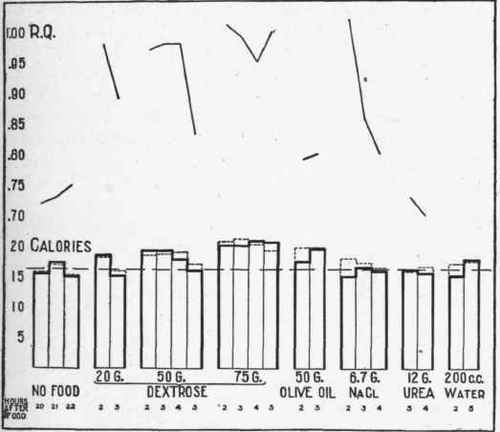
Fig. 20. - Illustrating the effect of the ingestion of glucose and fat and of water, urea, and salt solutions on the metabolism. Solid lines - metabolism in calories as calculated. Broken lines - metabolism in calories as found.
The experiments showed that the height of the metabolism was about the same whether 50 or 75 grams of glucose were given, only in the latter case the effect was more prolonged. This is in accord with Rubner's idea of "self-regulation," and also accords with the fact that the fermentative activity of living yeast cells is independent of the concentration of the sugar solution in which they live.1
What is the cause of the increased metabolism after the ingestion of glucose? In Fig. 20 it appears that the administration of 150 c.c. of water, either alone or containing 6.7 grams of sodium chlorid or 17 grams of urea, has no effect upon the heat production. Therefore neither osmotic exchanges nor increased kidney function, nor intestinal absorption can play any part in the increased heat production.
1 Rubner: "Sitzungsberichte der k. preussiscnen Akademie der Wissen-schaften," 1913, viii, 232.
These experiments led to further investigations1 to inquire into the cause of the rise in heat production after glucose ingestion. To this end various carbohydrates were given to the same dog in quantities of 50 grams. The fact that fructose exerted a more powerful influence on metabolism than glucose was confirmed. It was also shown that galactose oxidized with much greater difficulty in the dog than the other two monosaccharids, as evidenced by a lower metabolism and a lower respiratory quotient, and that lactose was not oxidized at all, and therefore caused no increase in the heat production. This latter fact must have been due to the absence of lactase from the intestine. These relations are shown in the following table:
Influence Of 50 Grams Of Various Carbohydrates Upon The Metabolism Of The Dog
Experiment No. | Sugar so Gm. | Average R.Q. | 2, 3, AND 4 Hours. |
Percentage of Increase Over Indirect Basal Metabolism. | |||
34,36 | Glucose........................ | 1.00 | 30 |
64, 66, 67 | Fructose....................... | 1.02 | 37 |
60,58 | Sucrose........................ | 1.02 | 34 |
67 | Galactose ........................... | 0.93 | 22 |
62 | Lactose........................ | 0.90 | 3 |
Weinland2 has shown that galactose does not form glycogen as readily as do glucose and fructose. From these facts it is most probable that galactose does not as readily dissociate into easily oxidizable molecules as do glucose and fructose.
1 Lusk: "Journal of Biological Chemistry," 1915, xx, 555. 2 Weinland: "Zeitschrift fur Biologie," 1899, xxxviii, 16 and 607.
By similar reasoning one may explain the difference in behavior between glucose and fructose. Glucose molecules require simple dehydration for conversion into glycogen and may in that fashion be removed from the tissue fluids. Fructose cannot be thus removed. It must first undergo chemical change, very likely by fragmentation into methyl-glyoxal with its three-carbon chain (formula on p. 265) before it can be synthesized into glycogen. These molecules being then in greater mass than similar molecules derived from ingested glucose would have been, it is easy to conceive that a higher metabolism would result on account of the greater plethora of oxidizable particles. F. G. Benedict1 states that the cause of the specific dynamic action of carbohydrates is the formation of acids, which act as stimuli. In support of this he cites an experiment by Benedict and Joslin,2 in which an increase of 30 per cent, in the heat production of a diabetic woman took place after the ingestion of 100 grams of fructose, notwithstanding the fact that the respiratory quotient before and after administration of the sugar was 0.69, indicating that none of the fructose was oxidized. Since fructose is transformed into glucose in the diabetic organism, Benedict interpreted this experiment as indicating the formation of acid intermediary products which stimulated metabolism.
Lusk has proved that when 12.5 grams of glycocoll, which are convertible into 10 grams of glucose, are given to a phlor-hizinized dog, the heat production is largely increased (see p. 244). Not so, however, with fructose. When 10 grams of fructose, which are convertible into 10 grams of glucose, are given to a phlorhizinized dog there is no increase whatever in metabolism. The chemical intermediates between fructose and glucose are without stimulating influence. This appears in the following experiment:
1 Benedict, F. G.: "Transactions of the XVth International Congress of Hygiene," 1912, ii, 394.
2 Benedict and Joslin: "Metabolism in Severe Diabetes," 1912, p. 69.
The Influence Of Fructose In Phlorhizin Glycosuria
Exp. No. | Conditions. | Hours. | Urinary D:N. | R.Q. | Nonprotein R.Q. | Calories. | |
Protein. | Total Indirect. | ||||||
81 | Eighth day fasting and phlorhizin.............. | 1 | 4.22 | 0.719 | 0.75 | 7.04 | 26.57 27.62 |
2 | 4.22 | 0.711 | 0.74 | 7.04 | |||
Average....... | 0.715 | 27.IO | |||||
Same after fructose 10 gm. | 2 | 7.31 | 0.697 | 0.72 | 6.08 | 25.80 | |
3 | 7.31 | 0.697 | 0.72 | 6.08 | 25.80 | ||
4 | 7.31 | 0.680 | 0.70 | O.08 | 24.78 | ||
Average....... | 0.692 | 25.46 | |||||
From the fact that the ingestion by a phlorhizinized dog of alanin, which certainly yields lactic or pyruvic acids in metabolism, causes a considerable rise in the heat production, (see p. 244), one may conclude that such acids are not intermediary metabolites in the reaction which converts fructose into glucose.
It may be added that if much acid be produced in carbohydrate metabolism, one would expect to find an increased quantity of ammonia in the urine during the oxidation of carbohydrate, just as ammonia elimination increases when lactic acid is formed in phosphorus-poisoning, but such an increase is not observed.1 Furthermore, if there were a considerable production of acid as a result of carbohydrate oxidation one would expect to find a fall in the quantity of carbon dioxid in the blood, such as occurs after giving meat, whereas unpublished experiments done by Dr. A. L. Meyer in the author's laboratory show that this is not the case - the quantity of carbon dioxid remains unchanged.
It is interesting to note that Freise2 found that when a surviving dog's liver is perfused with blood it yields 55 to 192 milligrams of carbon dioxid per minute per kilogram of substance. Addition of glucose, pyruvic acid, lactic acid, or glyceric acid increases the carbon dioxid 50 per cent., whereas galactose, glyoxylic, glycollic, and acetic acids were without influence.
1 Murlin and Lusk: "Journal of Biological Chemistry," 1915, xxii, 15. 2 Freise: "Biochemische Zeitschrift," 1913, liv, 474.
Continue to:


