Influence Of Ingestion Of Carbohydrate. Respiratory Metabolism. Part 3
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
Influence Of Ingestion Of Carbohydrate. Respiratory Metabolism. Part 3
It is, of course, known that the end-product of sugar metabolism, carbonic acid, is a stimulus to the respiratory center; but the end-product cannot be the cause of its own increased production for the following reasons: If methyl-glyoxal be an intermediate it may pass, on the one hand, back into glucose, and, on the other, forward into acetaldehyd the elimination of carbon dioxid.
C6H4206 - 2H20→ CH3.CO.CHO + H20→ CH3.CHO + C02 + H2
The reader is referred to p. 268, on which is described how by this process the transformation of carbohydrate into fat may occur. Now if, under these conditions, there is a great stimulation of metabolism, one would expect to find after giving an excess of carbohydrate that the heat production would be proportional to the amount of fat synthesized in the organism. This is emphatically not true. Neither the extra volume of carbon dioxid produced, which carries the respiratory quotient above unity, nor the extra metabolites involved in the reaction are effective in materially increasing the heat production (see p. 308).
It would seem that the real cause of the increased heat production after carbohydrate ingestion lay in the plethora of acetaldehyd molecules, which the cells, within the limits of the definite upper level imposed by self-regulation, were capable of utilizing. Above this level the acetaldehyd molecules are convertible into fat with little loss in the original energy content of the sugar from which they arise.
The subject may be approached from still another standpoint, combining the influence of carbohydrate with that of amino-acids and other food-stuffs. The following observations have been made by the author.1 When 50 grams of glucose were administered to a dog the heat production increased 30 per cent.; with 70 grams, the increase was 35 per cent. Twenty grams of glycocoll increased it 36 per cent., and the same amount of alanin, 32 per cent. Combined, 50 grams of glucose and 20 grams of glycocoll are the glucose equivalent of 66 grams, and yet when they were.given together the metabolism increased 56 per cent., an increase greater than 66 grams of glucose could have induced. Glucose and alanin in similar quantities are a glucose equivalent of 70 grams and caused an increase in heat production of 53 per cent. It is obvious that an increase in the quantity of glucose when this is given in large amounts scarcely affects metabolism; but that the chemical stimulus of amino-acids acting in conjunction with a plentiful supply of glucose results in a rise in heat production which is nearly the sum of the two individual influences acting separately. This points to a distinct difference between the cause of the specific dynamic action of glucose and that of alanin, which latter is convertible into lactic acid and eventually into glucose.
Lactic acid from alanin or glycollic acid from glycocoll may therefore raise the level of cell activity through direct stimulation; and if fragments of glucose metabolism be present in quantity, these may enter as .increased fuel to produce yet higher metabolism in the cells than the oxyacids would alone induce.
Also, when alcohol is given with glucose the metabolism rises above the level it would have attained had glucose been administered alone. The respiratory quotient falls, the cells oxidize both alcohol and the fragments of glucose metabolism, and produce almost as much extra heat as the sum of the quantities of heat which each material would have induced alone.
These experiments were extended by Murlin and Lusk,2 so that the influence of glycocoll, glucose, and fat when ingested severally and together could be analyzed.
1 Lusk: Loc. cit.
2 Murlin and Lusk: "Journal of Biological Chemistry," 1915, xxii, 15.
It was found that if glucose be ingested at the time of the highest fat metabolism, the heat production undergoes a second increase to the same extent which glucose alone would have increased metabolism. At this level of higher metabolism the respiratory quotient is 0.93 or 0.94, instead of unity, which it would have been if glucose had been given alone, hence glucose and fat were being oxidized together. The urinary ammonia fell following the ingestion of glucose four hours after fat ingestion, and this was true in spite of the combined oxidation of fat and carbohydrate. This does not suggest the presence of acid formation as the cause of the high metabolism.
When glucose and glycocoll are given together so that their molecules enter the circulation at the time of the height of fat absorption, the increase in metabolism is very nearly equal to the sum of the increases which each of the three materials would have induced alone.
The following table and accompanying chart (Fig. 21, p. 300) show these relations:
The Effect Of Glycocoll, Glucose, And Fat, Severally And Together Dog XIV. Second Series
Experiment Nos. | R.Q. | Cal. PER Hour. | No. OF Hours. | Increase Over Normal. | ||
15, 17, | 21 Basal....................... | 0.86 | 22.7 | 3 | Cal. | Pr. Ct. |
20 | Fat, 75 grams................ | 0.80 | 26.6 | 4* | 3.9 | 17 |
18 | Glycocoll, 20 grams.......... | 0.90 | 27.6 | 2 | 4.9 | 25 |
16, 19 | Glucose, 70 grams............ | 1.02 | 29.6 | 2 | 6.9 | 30 |
23 | Glucose, 50 grams + glycocoll, 20 grams.................. | 1.03 | 33.5 | 2 | 10.8 | 48 |
24 | Glucose, 50 grams + glycocoll, 20 grams, given four hours after fat, 75 grams . . . . . . | 1.02 | 37.3 | 2 | 14.6 | 64 |
* Hours 6, 7, 8, 9 after fat ingestion.
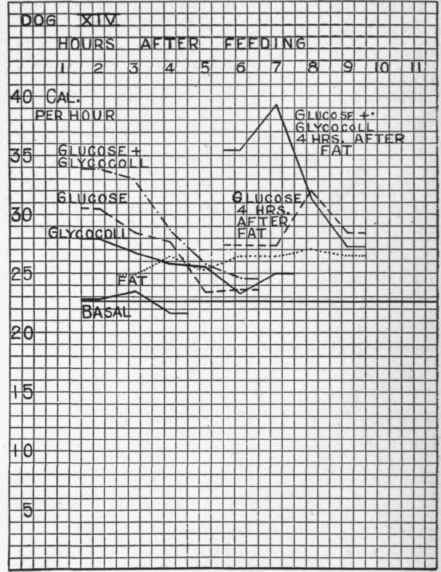
Fig. 21. - The effect of fat, of glycocoll, of glucose, of glucose + glycocoll, and of glucose + glycocoll + fat upon the heat production. (The "glucose four hours after fat" curve is atypical, is not similar to other experiments, and probably denotes a slow emptying of the stomach).
Continue to:


