The Oxidation Of Fats
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
The Oxidation Of Fats
In order to be able to understand the further fate of some of the deaminized remainders of the amino-acids, the method of oxidation of fatty acids must be understood. The experiments of Knoop3 are based on the fact that benzoic acid, C6H5COOH, when given to an animal pairs with glycocoll and appears as hippuric acid in the urine, whereas phenylacetic acid, C6H5CH2COOH, when given pairs in the same way and is eliminated as phenaceturic acid. Knoop found that whenever aromatic derivatives of the fatty acids were given to an animal one of these two forms always appeared in the urine; that if the side chain had an odd number of carbon atoms hippuric acid was always the end-result, and if there were an even number of atoms phenaceturic acid appeared as the final product.
The following substances were given:
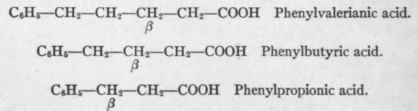
1 Kotake: "Zeitschrift fur physiologische Chemie," 1010, box, 409. 2 Ringer: "Journal of Biological Chemistry," 1913, xv, 145. 3 Knoop: "Hofmeister's Beitrage," 1905, vi, 150.
Since phenylvalerianic and phenylpropionic acids both yielded hippuric acid and phenylbutyric did not, it was evident that the last named was not an intermediary product between the first two. Knoop, therefore, concluded that in the oxidation of fats the β-carbon atom was oxidized and that two carbon atoms dropped from the chain together. This view was supported by Dakin's1 discovery that when phenylpropionic acid was given in large amounts phenyl-β-oxy-propionic acid, C6H5 - CHOH - CH2 - COOH, was detected in the urine. Corroborative evidence is further found in the fact that when body fat or food fat, both of which always contain an even number of carbon atoms, are metabolized in the diabetic, the end-product is always β-oxybutyric acid, CH3CHOHCH2-COOH, which one would expect in terms of the theory.
It is interesting to note the results of giving the following three substances:
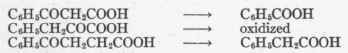
Phenyl-β-keto-propionic acid is oxidized on the β-carbon atom to benzoic acid. Phenyl-α-keto-propionic acid (phenyl-pyruvic acid), as stated, is completely oxidized in the organism, while phenyl-γ-keto-butyric acid undergoes reduction of its γ-carbon and oxidation of its β-carbon and is eliminated in the urine as phenyl-acetic acid. Here reduction and oxidation play alternately upon the same molecule.
The oxidation of unsaturated fatty acids, or such as have a double linkage between two carbon atoms, follows the same laws as the oxidation of saturated fatty acids. Thus, Erdmann and Marchand2 found that if cinnamic acid, C6H5.CH:CH.COOH, be given to an animal, benzoic acid appears in the urine. Dakin3 administered the material in large doses to cats and found the intermediary oxidation product, phenyl-β-oxy-pro-pionic acid, C6H5.CHOH.CH2COOH, in the urine.
1 Dakin: "Journal of Biological Chemistry," 1909, vi, 203. 2 Erdmann and Marchand: "Liebig's Annalen," 1842, xliv, 344. 3 Dakin: "Oxidations and Reductions in the Animal Body," 1912, p. 36.
This preliminary discussion has shown that amino-acids are oxidized at the α-position which is the point of attachment of the NH2 group, and are converted into oxy- or keto-acids and then into acids having one less carbon atom. After this the organic acid becomes subject to the laws of /3-oxidation, under which a fatty acid is oxidized on its β-carbon atom, oxy-and keto-acids being first formed, and then there is cleavage of two carbon atoms with the formation of an acid which contains two less carbon radicles than before. Frequently β-oxybutyric acid is an intermediary product of this oxidation, just as happens in the case of fatty acids. In other cases in which the product contains three atoms of carbon, the end-product may be converted into glucose in the organism. Ringer1 has demonstrated that in the /3-oxidation of fatty acids having uneven numbers of carbon atoms sugar is formed from them in the diabetic organism in proportion to the power to produce propionic acid, CH3.CH2.COOH. This might form β-lactic acid, CH2OH.CH2COOH, which, in turn, might be converted into glucose.
Experiments have shown that the glucose-forming amino-acids include glycocoll, alanin, prolin, aspartic and glutamic acids, serin, cystin, and arginin. Some of the other amino-acids yield β-oxybutyric acid as an intermediary product.
It seems desirable at this point to enter into the more intimate details of the life-history of the different amino-acids. Though the general energy metabolism may be understood without this knowledge, yet the finer comprehension of the subject cannot be otherwise obtained.
Continue to:


