The Process Of Deamination
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
The Process Of Deamination
The nature of the attack of the living cell upon the NH2 group of the amino-acids has been the subject of much investigation. The process was at first thought to be one of simple hydrolysis, as follows:
R-CHNH2-COOH + HOH = R-CHOH-COOH + NH3
After this fashion glycocoll, CH2NH2 - COOH, would become glycollic acid, CH2OH - COOH; alanin, CH3 - CHNH2 - COOH, would become lactic acid, CH3 - CHOH - COOH, and so forth.
It was Otto Neubauer,2 in the laboratories of the second medical clinic of the University of Munich, who first showed that the process of deamination- might be one oxidation and not, hydrolysis. This process of oxidative deamination is represented in the following formula-: - .
R-CHNH2- COOH + O = R-CO-COOH + NH3
From glycocoll, CH2NH2 - COOH, one would thus obtain gly-oxylic acid, CHO - COOH; and from alanin, CH3 - CHNH2 - COOH, pyruvic acid, CH3 - CO - COOH.
That this method of oxidation is actually possible in the organism was evident when Neubauer gave phenyl-glycocoll to a dog and found phenyl-glyoxylic acid as well as mandelic acid in the urine.
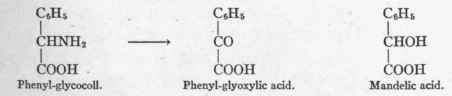
1 For excellent reviews, see Dakin: "Oxidations and Reductions in the Animal Body," Longmans, Green and Co., 1912; Underhill, "The Physiology of the Amino-Acids," Yale University Press, 1915.
2 Neubauer: "Deutsches Archiv fur klinische Medizin," 1909, xcv, 211.
Further evidence was obtained by Neubauer through the medium of a rare anomaly of human metabolism called alcaptonuria (see p. 196). In this disease tyrosin and phenyl-alanin are not oxidized to their usual end-products, but are eliminated in the urine as homogentisic acid. The transformation of phenyl-alanin and tyrosin into homogentisic acid is believed to follow the scheme presented below:
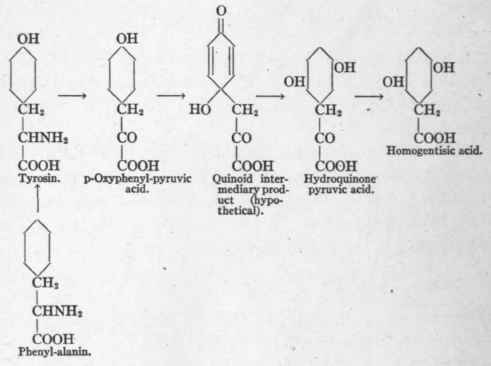
It will be noted that the alanin radicle, CH2- CHNH2- COOH, is represented as undergoing oxidative deamination, being converted into pyruvic acid. Neubauer drew this conclusion from the fact that if phenyl-alanin, tyrosin, or p-oxy-phenyl-pyruvic acid were given to the alkaptonuric patient they all appeared in the urine as homogentisic acid, whereas when p-oxy-phenyl-lactic acid was given there was no increase in the homogentisic acid excretion whatever. Consequently it could not have been an intermediary product in the metabolism of tyrosin. Neubauer, therefore, concluded that the primary pathway of deamination was oxidative and not hydrolytic. Later he1 presented the following formula as indicating the probable reaction of oxidative deamination:

Alanin, for example, would follow this pathway:

Examination of the formula given for the conversion of phenyl-alanin into homogentisic acid shows that the alanin radicle of phenyl-alanin is converted into an acetic acid radicle in homogentisic acid. The question arises whether the first step in the destruction of phenyl-alanin might not be the loss of its acid group by CO2 cleavage, as indeed happens when it is acted upon by bacteria,2 and as is usual in the transformation of cystein into taurin.

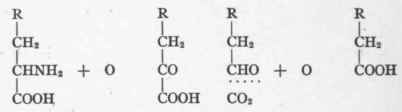
As phenyl-ethyl-amin is poisonous, its first oxidation product, phenyl-ethyl-alcohol, was given by Neubauer to the alkaptonuria but without increasing the quantity of homogentisic acid. It appeared in the urine as phenyl-acetic acid (paired with glycocoll). This indicates that oxidative deamination takes place in the metabolism of phenyl-alanin before CO2 is split from the acid radicle. The CO2 cleavage follows deamination, as appears in the formula given for the transformation of hydroquinone-pyruvic acid into homo-gentisic acid. It follows from this that after the oxidative deamination of an amino-acid, the deaminized remainder may be converted into an acid containing one less carbon atom, as follows:
1 Neubauer and Fromherz: "Zeitschrift fur physiologische Chemie," 1910, lxx, 326.
2 Spiro: "Hofmeister's Beitrage," 1902, i, 347.
If after producing the aldehyde by C02 cleavage, reduction prevails instead of oxidation, then an alcohol is formed instead of an acid. This may be illustrated by the work of F. Ehrlich,1 who found that when yeast acted on tyrosin the end-product was p-oxy-phenyl-ethyl-alcohol, OH - C6H4 - CH2-CH2OH. Neubauer and Fromherz, continuing their theoretic researches, discovered that yeast acting on p-oxy-phenyl-pyruvic acid yields this same ethyl-alcohol derivative, while p-oxy-phenyl-lactic acid does not give it. Para-oxy-phenyl-pyruvic acid may, therefore, be transformed as follows:
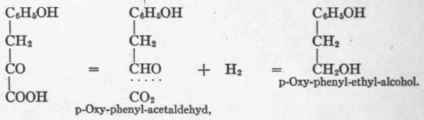
Although p-oxy-phenyl-lactic acid is not acted on by yeast, yet it also appears as a product when yeast acts on p-oxy-phenyl-pyruvic acid. Hence pyruvic acid may be reduced, with the formation of lactic acid.
1 Ehrlich, F.: "Ber. d. d. chem. Ges.," 1907, xl, 1047.
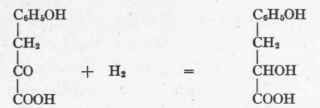
In conformity with this stands the observation of P. Mayer,1 who found that if pyruvic acid were administered to a rabbit lactic acid appeared in the urine.
Although it appears certain that oxidative deamination is the principal method of attack upon the amino group of the aromatic acids, yet direct hydrolytic deamination has been noted for them, and it may play a considerable role in the metabolism of the amino-acids of the aliphatic series as well. Neubauer finds the laevo-component of p-oxy-phenyl-lactic acid in the urine of a patient suffering from cirrhosis of the liver. Since the dextro-component is always eliminated in the human being whenever it is formed by reduction of p-oxy-phenyl-pyruvic acid within the organism, it follows that this latter substance could not have been the intermediary one, but that the 1-compound was formed by the direct hydrolytic cleavage of 1-tyrosin.
These reactions give one an insight into oxidations, reductions, hydrolyses, and cleavages, which are of constant occurrence as the result of vital activities. One may summarize all these possible biologic variations in the following scheme, using alanin as a typical amino-acid:

1 Mayer, P.: "Biochemische Zeitschrift," 1912, xl, 441.
It has been noted by Kotake1 that although p-oxy-phenyl-pyruvic acid is completely oxidized when administered to a rabbit, p-oxy-phenyl-lactic acid remains almost untouched and appears in the urine. The fact, however, that lactic acid and alanin pass over into sugar much more readily than does pyruvic acid leads Ringer2 to believe that the metabolism of alanin probably follows the path of hydrolytic deamination into lactic acid rather than that of oxidative deamination into pyruvic acid.
The reader should realize that there are many possible pathways in metabolism, and the above presentation may be regarded as suggestive rather than literally exact.
Continue to:


