Arginin, Nh2.CNHNH.CH2CH2CH2.CHNH2.COOH
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
Arginin, Nh2.CNHNH.CH2CH2CH2.CHNH2.COOH
Present in most proteins. Probably three carbon atoms form glucose.
Kossel and Dakin6 found that liver but not muscle contained an enzyme capable of splitting d-arginin into urea and ornithin, the only reaction by which urea is obtained as a simple cleavage product of an amino-acid. Dakin1 gave both arginin and ornithin to a phlorhizinized dog, and witnessed a sufficient elimination of extra glucose to account for three of the carbon atoms in these substances. Since three carbon atoms of succinic acid enter into the formation of glucose, and succinic acid appears to be a possible product of the metabolism of ornithin, one may assume that this might represent the pathway into sugar. These formulae may thus be portrayed:
1 Dakin: Loc. cit.
2 Ringer, Frankel, and Jonas: Loc. cit.
3 Ringer: "Journal of Biological Chemistry," 1912, xii, 223.
4 Literature, v. Furth: "Probleme der physiologischen und pathologischen Chemie," 1913, Bd. II, p. 117.
5 Kossel and Dakin: "Zeitschrift fur physiologische Chemie," 1904, xli, 321; 1904, xlii, 183.
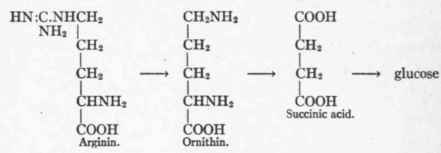
Another possible pathway would be the oxidation of the δ-C atom of ornithin to COOH, with the production of glutamic acid, which would then break up with sugar formation (see p. 201).
Bacteria in the intestine may convert ornithin into putresin, NH2 - C4H8 - NH2, through the cleavage of CO2 (see p. 203).
Arginin is the only amino-acid containing the guanidin nucleus, which is also found in creatin. However, attempts to associate the origin of creatin from arginin have proved fruitless. Jaffe2 gave nitrate of arginin subcutaneously to a rabbit, but found no change in the amount of creatin in the urine or muscle. According to the known laws of oxidation of amino-acids, guanidin acetic acid, NH2.CNH.NH.CH2.COOH, might readily be an oxidation product of arginin. When Jaffe gave this substance to a rabbit he found that a methyl group was added and it was in part eliminated in the urine as creatin, NH2.CNH.NCH3.CH2.COOH. The proof of the origin of guanidin acetic acid in the organism is, however, lacking. Arginase does not effect the cleavage of creatin or guanidin acetic acid1 (see also p. 200).
1 Dakin: Loc. cit.
2 Jaffe: "Zeitschrift fur physiologische Chemie," 1906, xlviii, 430.
Present in most proteins. Does not produce glucose, and there is no clear evidence that it produces β-oxybutyric acid.
When histidin is given to dogs it is oxidized and urea formed from it appears in the urine.2 When it is given to phlor-hizinized dogs Dakin3 finds no clear-cut evidence that it is converted either into glucose or β-oxy butyric acid.
Histidin gives Ehrlich's diazo-reaction, and is probably the chromogen within urochrom.4 proteins. Three carbon atoms enter into the formation of glucose.
Dakin5 gave prolin to a phlorhizinized dog and found extra glucose eliminated to the extent of three of the five carbon atoms contained in prolin. Ringer suggests that the intermediary product may be succinic acid, but lactic acid or glyceric acid are also possible. The metabolism probably follows the same lines as does that of glutamic acid (see p. 202).
Continue to:


