Tryptophan (Formula Below)
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
Tryptophan (Formula Below)
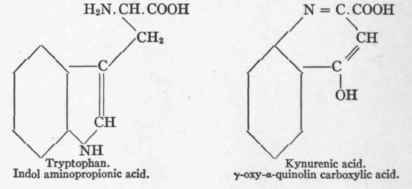
Present in animal proteins except gelatin. Absent in zein. Produces neither glucose nor β-oxybutyric acid, but is convertible into kynurenic acid.
Dakin6 could find no certain increase in sugar or /3-oxybutyric acid excretion after giving 14.5 grams of tryptophan to a phlorhizinized dog.
Ellinger7 discovered that the production of kynurenic acid, which had long been known as a component of dog's urine, was largely increased after giving tryptophan. Mendel and Jackson1 found that the kynurenic acid elimination in dogs varied directly with the protein metabolism, but was not derived from gelatin metabolism. Ellinger also gave tryptophan to a rabbit, whose urine normally contains no kynurenic acid, and found kynurenic acid in the urine. Rabbits however, normally oxidize kynurenic acid when ingested in small amounts. He reaches the conclusion that animals in general may produce kynurenic acid, and that this is usually readily oxidized except in the organism of the dog, where it is only partly destroyed, and therefore appears in the urine.
1 Dakin: "Journal of Biological Chemistry," 1907, iii, 435.
2 Abderhalden and Einbeck: "Zeitschrift fur physiologische Chemie," 1909, lxii, 322; 1910, lxviii, 395.
3 Dakin: "Journal of Biological Chemistry," 1913, xiv, 328.
4 von Furth: "Probleme der physiologischen und pathologischen Chemie," 1913, ii, 605.
5 Dakin: "Journal of Biological Chemistry," 1912-13, xiii, 513.
6 Dakin: Ibid., 1913, xiv, 321.
7 Ellinger: "Zeitschrift fur physiologische Chemie," 1904, xliii, 325.
Hopkins and Cole2 first isolated tryptophan in a state of purity and determined its formula. Miss Homer3 determined the exact formula of kynurenic acid. Ellinger4 thus presents the transformation of tryptophan into kynurenic acid:
Intestinal bacteria act upon the propionic acid radicle of tryptophan and convert it into skatol or indol:

1 Mendel and Jackson: "American Journal of Physiology," 1898-99, ii, 1. 2Hopkins and Cole: "Journal of Physiology," 1901-02, xxvii, 418. 3Homer: "Journal of Biological Chemistry," 1914, xvii, 509. 4 Ellinger and Matsuoka: "Zeitschrift fur physiologische Chemie," 1914, xci, 45.
Indol and skatol, together with phenol, C6H5.OH, and p-cresol, CH3.C6H4.OH, the products of bacterial putrefaction of phenylalanin and tyrosin, are conjugated with sulphuric acid in the liver and are eliminated as ethereal sulphates in the urine.
Continue to:


