Hot Water Supply - Water Heating Apparatus Properties Of Heat
Description
This section is from the book "Principles And Practice Of Plumbing", by John Joseph Cosgrove. Also available from Amazon: Principles and Practice of Plumbing.
Hot Water Supply - Water Heating Apparatus Properties Of Heat
Transfer Of Heat
When two bodies of different temperatures are near each other a transfer of heat takes place from the hotter to the colder body. This tendency towards maintaining an equilibrium of temperature is universal and the transfer of heat may take place in any of three ways, by conduction, by convection or by radiation.
Conduction is the progressive movement of heat through a substance without perceptible movement of the molecules; if one end of a poker is held in a fire, the other end will become heated by conduction. Water in a waterback or vessel becomes heated from the flames and hot gases of a fire by conduction of heat through the metal walls of the waterback or vessel.
Convection is the transfer of heat by movement or circulation of the molecules of the substance to be heated. Water in a vessel placed on a stove is heated by local circulation of the water. Fluids, such as air or water, can be heated only by convection. This is due to the fact that when heat is applied to a fluid, the particles in contact with the heat expand in bulk, consequently become lighter in weight and are replaced by colder and denser particles.
Radiation is the transmission of heat from a warm body to one of lower temperature. For example, the earth is warmed by radiation from the sun. Radiant heat does not heat the air through which it passes; it travels directly and in straight lines until intercepted and reflected or absorbed by some other body. The cooler body will reflect or absorb all the heat rays it intercepts and the sum of the absorption and reflection equals the total of the intercepted rays.
Absorption and radiation are equal and opposite. The better the absorptive power of a substance the better radiating material it would make. Lampblack, which has absorbing and radiating powers rated at 100, is taken as the standard of comparison. In proportion as the reflecting power of a substance diminishes, its power to absorb or radiate heat increases. The absorbing, radiating and reflecting capacity of various substances are given in Table XLIV.
Table XLIV - Absorption And Radiation
Substance | Powers | |
Radiating or Absorbing | Reflecting | |
Lampblack ............................. | 100 | 0 |
Water.......... ........................ | 100 | 0 |
Carbonate of lead........................ | 100 | 0 |
Writing paper........................... | 98 | 2 |
Marble | 93 to 98 | 7 to 2 |
Isinglass | 91 | 9 |
Ordinary glass | 90 | 10 |
Ice..................................... | 85 | 15 |
Cast iron................................ | 25 | 75 |
Wrought iron, polished | 23 | 77 |
Steel, polished | 17 | 83 |
Tin......................-................ | 15 | 85 |
Brass, cast, dead polished................ | 11 | 89 |
Brass, hammered, dead polished.......... | 9 | 91 |
Brass, cast, bright polished.............. | 7 | 93 |
Brass, hammered, bright polished........ | 7 | 93 |
Copper, varnished...................... | 14 | 86 |
Copper deposited on iron................. | 7 | 93 |
Copper, hammered or cast............... | 7 | 93 |
Measurement Of Heat
The amount of heat transmitted to water is measured by the British Thermal Unit usually abbreviated B. T. U. A B. T. U. is the quantity of heat required to raise the temperature of one pound of water from 62 to 63 degrees Fahr. In practice it is taken as the quantity of heat required to raise one pound of water 1 degree Fahr.
Measurement Of Temperature
The temperature of water is measured by a mercury thermometer. For measuring water temperatures, thermometers, Fig. 116, should have a scale ranging from 60 degrees Fahr. to 270 degrees Fahr., and should be so constructed that when screwed into a fitting the mercury bulb, a, will project into the pipe and thus be in contact with the hot water.
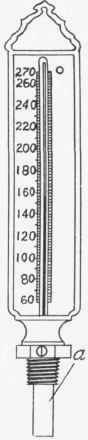
Fig. 116
Transmission Of Heat
The quantity of heat transmitted to water through a vessel or tube depends on the difference in temperature between the heating medium and the absorbing water, the thickness of the walls of the vessel or tube, and the material of which it is made. All other conditions being equal, copper pipes will transmit 50 per cent, more heat than iron pipes, and cast iron surfaces will transmit about 60 per cent, less than an equal area of iron pipe surface. The relative transmission of heat for different metals is shown in Table XLV.
Table XLV - Transmission Of Heat
Experimenters | Character of Surface | Steam condensed per square foot per degree difference of temperature per hour | Heat transmitted per square foot per degree difference of temperature per hour | Remarks | ||
Heating Pounds | Evaporating Pounds | Heating B.T.U. | Evaporating B.T.U. | |||
Laurens | Copper coils 2 Copper coils Copper coil | .292 .268 | .981 1.20 1.26 | 315 280 | 974 1120 1200 | |
Perkins | Iron coil | .... | .24 | .... | 215 | 100 lbs. Pressure |
Perkins | Iron coil | .... | .22 | .... | 208.2 | 10 lbs. Pressure |
Box | Iron tube | .235 | * • • • | 230 | ||
Box | Iron tube | .196 | 230 | |||
Box | Iron tube | .206 | 207 | |||
Havrez | Cast iron boiler | .077 | .105 | 82 | 100 | |
Kent's Pocketbook
From the above table of experiments, the following table of average heat units transmitted through various substances is adduced. The table is based on the assumption that the outer surface is clean and free from soot or ashes, and that the inner surface is free from incrustations of lime or other substances.
Table XLVI - Transmission Of Heat
Materials | Heat transmitted per square foot of heating surface each hour for each degree Fahr. difference between the heating medium and the water |
Copper plate ...................... | 275 B. T. U. |
Copper pipe | 300 B. T. U. |
Wrought iron or steel pipe or surface | 200 B. T. U. |
Cast iron surface | 80 B. T. U. |
Continue to:


