Ballistics. Part 8
Description
This section is from "The Encyclopaedia Britannica". Also available from Amazon: Great Books of the Western World (60 Volumes).
Ballistics. Part 8
In Sir Andrew Noble's researches a number of plugs were inserted in the side of the experimental gun, reaching to the bore and carrying crusher-gauges, and also chronographic appliances which registered the passage of the shot in the same manner as the electric screens in Bashforth's experiments; thence the velocity and energy of the shot was inferred, to serve as an independent control of the crusher-gauge records (figs. 4 and 5).
As a preliminary step to the determination of the pressure in the bore of a gun, it is desirable to measure the pressure obtained by exploding a charge of powder in a closed vessel, varying the weight of the charge and thereby the density of the powder-gas.
The earliest experiments of this nature are due to Benjamin Robins in 1743 and Count Rumford in 1792; and their method has been revived by Dr Kellner, War Department chemist, who employed the steel spheres of bicycle ball-bearings as safety-valves, loaded to register the pressure at which the powder-gas will blow off, and thereby check the indications of the crusher-gauge (Proc. R.S., March 1895).
Chevalier d'Arcy, 1760. also experimented on the pressure of powder and the velocity of the bullet in a musket barrel; this he accomplished by shortening the barrel successively, and measuring the velocity obtained by the ballistic pendulum; thus reversing Noble's procedure of gradually lengthening the gun.
But the most modern results employed with gunpowder are based on the experiments of Noble and Abel (Phil. Trans., 1875-1880-1892-1894 and following years).
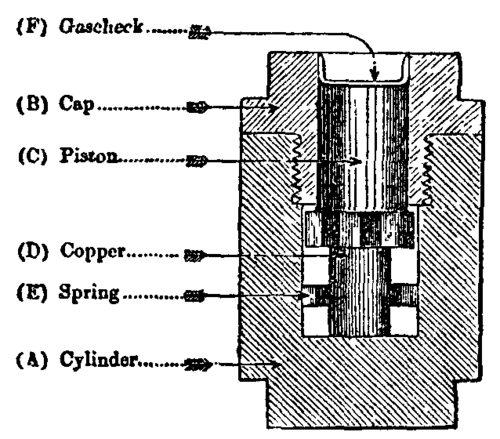 Fig. 6.
Fig. 6.
A charge of powder, or other explosive, of varying weight P lb, is fired in an explosion-chamber (fig. 7, scale about 1/5) of which the volume C, cub. in., is known accurately, and the pressure p, tons per sq. in., was recorded by a crusher-gauge (fig. 6).
The result is plotted in figs. 8 and 9, in a curve showing the relation between p and D the gravimetric density, which is the specific gravity of the P lb of powder when filling the volume C, cub. in., in a state of gas; or between p and v, the reciprocal of D, which may be called the gravimetric volume (G.V.), being the ratio of the volume of the gas to the volume of an equal weight of water.
 Fig. 7. Explosion Vessel.
Fig. 7. Explosion Vessel.
The results are also embodied in the following Table; -
Table 1.
G.D. | G.V. | Pressure in Tons per sq. in. | |
Pebble Powder. | Cordite. | ||
0.05 | 20.00 | 0.855 | 3.00 |
6 | 16.66 | 1.00 | 3.80 |
8 | 12.50 | 1.36 | 5.40 |
0.10 | 10.00 | 1.76 | 7.10 |
12 | 8.33 | 2.06 | 8.70 |
14 | 7.14 | 2.53 | 10.50 |
15 | 6.66 | 2.73 | 11.36 |
16 | 6.25 | 2.96 | 12.30 |
18 | 5.55 | 3.33 | 14.20 |
20 | 5.00 | 3.77 | 16.00 |
22 | 4.54 | 4.26 | 17.90 |
24 | 4.17 | 4.66 | 19.80 |
25 | 4.00 | 4.88 | 20.63 |
26 | 3.84 | 5.10 | 21.75 |
30 | 3.33 | 6.07 | 26.00 |
35 | 2.85 | 7.35 | 31.00 |
40 | 2.50 | 8.73 | 36.53 |
45 | 2.22 | 10.23 | 42.20 |
50 | 2.00 | 11.25 | 48.66 |
55 | 1.81 | 13.62 | 55.86 |
60 | 1.66 | 15.55 | 63.33 |
The term gravimetric density (G.D.) is peculiar to artillerists; it is required to distinguish between the specific gravity (S.G.) of the powder filling a given volume in a state of gas, and the specific gravity of the separate solid grain or cord of powder.
Thus, for instance, a lump of solid lead of given S.G., when formed into a charge of lead shot composed of equal spherules closely packed, will have a G.D. such that
| (4) | G.D. of charge of lead shot | = | 1 | π√2 = 0.7403; |
| S.G. of lump of solid lead | 6 |
while in the case of a bundle of cylindrical sticks of cordite,
| (5) | G.D. of charge of cordite | = | 1 | π√3 = 0.9067. |
| S.G. of stick of cordite | 6 |
At the standard temperature of 62° F. the volume of the gallon of 10 lb of water is 277.3 cub. in.; or otherwise, 1 cub. ft. or 1728 cub. in. of water at this temperature weighs 62.35 lb, and therefore 1 lb of water bulks 1728 ÷ 62.35 = 27.73 cub. in.
 Fig. 8.
Fig. 8.
Thus if a charge of P lb of powder is placed in a chamber of volume C cub. in., the
(6) G.D.= 27.73P/C, G.V. = C/27.73 P.
Sometimes the factor 27.68 is employed, corresponding to a density of water of about 62.4 lb per cub. ft., and a temperature 12° C., or 54° F.
With metric units, measuring P in kg., and C in litres, the G.D. = P/C, G.V. = C/P, no factor being required.
From the Table I., or by quadrature of the curve in fig. 9, the work E in foot-tons realized by the expansion of 1 lb of the powder from one gravimetric volume to another is inferred; for if the average pressure is p tons per sq. in., while the gravimetric volume changes from v - ½δv to v + ½δv, a change of volume of 27.73δv cub. in., the work done is 27.73pδv inch-tons, or
(7) δE = 2.31 pδv foot-tons;
and the differences δE being calculated from the observed values of p, a summation, as in the ballistic tables, would give E in a tabular form, and conversely from a table of E in terms of v, we can infer the value of p.
On drawing off a little of the gas from the explosion vessel it was found that a gramme of cordite-gas at 0° C. and standard atmospheric pressure occupied 700 ccs., while the same gas compressed into 5 ccs. at the temperature of explosion had a pressure of 16 tons per sq. in., or 16 × 2240 / 14.7 = 2440 atmospheres, of 14.7 lb per sq. in.; one ton per sq. in. being in round numbers 150 atmospheres.
The absolute centigrade temperature T is thence inferred from the gas equation
(8) R = pv / T = pv/273,
which, with p = 2440, v = 5, p = 1, v = 700, makes T = 4758, a temperature of 4485° C. or 8105° F.
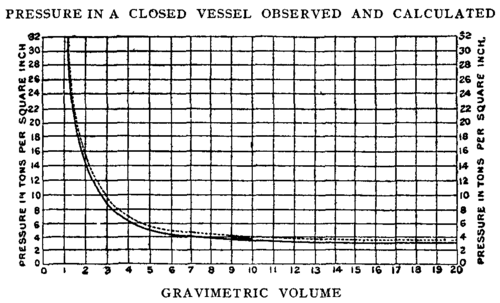 Fig. 9.
Fig. 9.
In the heading of the 6-in. range table we find the description of the charge.
Charge: weight 13 lb 4 oz.; gravimetric density 55.01/0.504; nature, cordite, size 30.
So that P = 13.25, the G.D. = 0.504, the upper figure 55.01 denoting the specific volume of the charge measured in cubic inches per lb, filling the chamber in a state of gas, the product of the two numbers 55.01 and 0.504 being 27.73; and the chamber capacity C = 13.25 × 55.01 = 730 cub. in., equivalent to 25.8 in. or 2.15 ft. length of bore, now called the equivalent length of the chamber (E.L.C.).
If the shot was not free to move, the closed chamber pressure due to the explosion of the charge at this G.D. (= 0.5) would be nearly 49 tons per sq. in., much too great to be safe.
Continue to:


