Heat And Cold
Description
This section is from the book "The Corner Cupboard; Or, Facts For Everybody", by Robert Kemp Philp. Also available from Amazon: The Corner Cupboard; or Facts for Everybody.
Heat And Cold
Heat And Cold. Cold is the absence of heat; and heat is a peculiar influence of which we can only judge by its effect.
There are two states in which we find heat. In the one case we recognize it by the touch, in the other we cannot find it out by such means. The air has a very large quantity of heat hidden in it, even when it seems coolest to you. It contains so much heat, even in the depth of winter, that the quantity contained in a few square inches, if squeezed out, is sufficient to light a piece of tinder. We have drawn here a figure of a solid square, or cube, that we may be fully understood Each face has four edges, as in fig. 1, and there are six faces to the solid square, as in fig. 2. Now if you had a vessel which would hold about twelve square inches or cubes of air, and could so manage as to squeeze it into the size of one square inch, so much heat would be given out that a piece of tinder would be set on fire by it.
If you procure a piece of thick iron wire, and hammer the end of it on an anvil or hard stone, you will find that after a few blows it has become very hot. You have compressed the particles of the iron, and the latent, or hidden, heat, has become sensible to the touch. The heat kept the particles or atoms of iron apart from each other; but when you have hammered them close, your hammering will produce no more heat. There is only a certain quantity of juice to be squeezed out of an orange, and so the quantity of heat in the iron is limited. You have learned by this experiment, which any one may perform with a piece of soft iron wire, that heat has a tendency to separate the particles of which bodies are composed, and make them larger. This separation is called expansion. A bar of iron, when heated, becomes longer; and hence, if the iron pieces or plates upon which the wheels of the railway engines run were to be placed in winter with their ends close against each other, they would be so expanded by the warmth of the sun in summer, that they would become crooked, and the line would be unsafe to travel upon. This expansion of bodies by heat is taken advantage of by persons who wish to measure sensible heat. Thermometers are constructed of bodies which are readily expansible by heat. Those commonly in use are made of quicksilver shut up in glass. The glass tube is mounted on a piece of bone or ivory, which bears the marks of degrees, so that the amount of expansion or contraction of the quicksilver can be stated or registered. There are various forms of heat measurers or thermometers, and I will explain to you how to construct a simple one. Take a wide-mouthed bottle, pour into it a teaspoonful of ink, and two table-spoonfuls of water, and set it upon the table. Having procured a glass tube, which has been blown into a bulb at one end, warm it thoroughly before the fire, and then rapidly place it in the bottle, so that the open end is plunged into the ink and water at the bottom. In a second cr two the air in the round bulb of the tube will begin to contract, and the inky water will ascend the tube, and perhaps rush into the bulb. When all upward motion in the water has ceased, place your hand upon the bulb, and you will then, by warming the air inside, cause it to expand, and the water in the tube will again descend. Pure water would answer as well as the mixture of ink, but the latter is more easily seen as it ascends and descends the tube.



If you place the bulb and bottle near the fire. the air in the bulb will be expanded still further. Your thermometer just shows that heat has been taken from the tire and combined with the air in the bulb. But how was the heat eonveyed from the fire to the bulb? By what means was it coinmu-nicated?
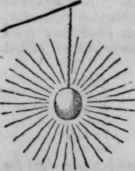
Heat is conveyed in two different manners. A heated globe of iron, if suspended in the air, would send out rays, or straight lines of heat, in every direction, as in this diagram. When heat is given off and conveyed to another body by rays in this manner, it is said to be by radiation. To illustrate the other mode in which heat travels, hold the end of a piece of iron wire, of three inches in length, in the flame of a small taper. The radiated heat from the flame you will scareely feel, but the heat will be speedily conducted to your fingers along the wire, which will become so hot that you will scarcely be able to hold it. When heat is thus conveyed by a metal or other solid body from a fire or flame to any other body, it is said to be by conduction. Metals conduct heat rapidly, and hence metal tools which require to be used while hot have the handles covered with wood, which does not allow the warmth to pass from the iron to the hand of the workman. You could hold a piece of wood of much shorter length than the wire, in the taper's flame, and feel no inconvenience. These circumstances prove that wood docs not conduct or convey heat at as rapidly as iron; it is therefore said to be a bad conductor ; in other words, its power of transmitting warmth is small. Woollen cloth is a still worse conductor; and for that reason, when the flat iron is used by the laundress, she protects her fingers with a piece of folded flannel, which does not allow the heat from the handle of the iron to burn her hand.

You have seen how the air is expanded by heat, and contracted by cold ; it is important, morever, to understand, that in a cubic inch of cold air there are more particles than in a cubic inch of heated air. The figure A represents a cold cubic inch of air, which we will suppose contains sixteen atoms or particles, of air. If to this be applied a very small quantity of heat, it would expand to the size of B, without any increase of the number of particles, or in its weight. A cubic inch (as indicated by the dotted lines C and D), would then only contain nine atoms or particles of air.

If the cold cubic inch weighed sixteen grains, the hot cubic inch would only weigh nine. If bodies are expanded by heat, they contain fewer particles in a given space; if they are contracted by the removal of that heat, they contain more particles in the same space. Some of these particles would be oxygen and some nitrogen, always bearing the same proportion.
Continue to:


