Introduction. Part 7
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Introduction. Part 7
Relation of Constant C to Molecular Weight and Constitution
1.In any homologous series, or any series of closely related substances, except the alcohols, acids, phenols, the lower esters and perhaps some others, the higher the molecular weight the lower is the constant. Examples: - The normal paraffins; methyl and ethyl ethers; toluene and meta-xylene; acetone and benzophenone; benzene, naphthalene and anthracene.
2. Iso-compounds have higher values than their normal isomerides, and if there are two iso-groups the value is still higher. Examples: -Isopentane and normal pentane; di-isobutyl and n-octane; methyl isobutyrate and methyl butyrate.
3. When hydrogen is replaced by a halogen, the value is lowered.
Examples : - Benzene and a mono-derivative ; naphthalene and bromo-naphthalene.
4. By replacing one halogen by another no change is usually produced. Examples: - The four halogen derivatives of benzene.
5. All compounds containing a hydroxyl group - alcohols, phenols, water, acids - have very low values. But the influence of the hydroxyl group in lowering the constant diminishes as the complexity of the rest of the molecule increases. Thus, with methyl, ethyl, and propyl alcohols the constants must be as much as 0.000035 lower than those of the corresponding hydrocarbons, but with amyl alcohol it is only 0.000029, and with phenol only 0.000015 lower. On the other hand, the constant tends in general to be lowered as the molecular complexity increases, and these two factors, acting in opposite directions, neutralise each other more or less completely ; thus, in the case of the alcohols at any rate, there is apparently no relation between the values of the constant and the molecular weight.
6. The esters - formed from alcohols and acids - have rather low values, and here again the constant is nearly independent of the molecular weight.
Modifications Of The Still
For ordinary laboratory purposes a round-bottomed glass flask is the most convenient form of still, but if a large quantity of liquid has to be distilled, especially when it is very inflammable, it is safer to employ a metal vessel. Metal vessels are generally made use of on the large scale.
Any alteration in the shape of the still as a rule is merely a matter of convenience and does not call for special mention.
Modifications of the still-head are of great importance and will be considered later in Chapters X. to XIII.
Modifications Of The Condenser
For liquids boiling above the ordinary temperature, but below about 170°, the straight Liebig's condenser is usually employed, but various more compact and efficient forms of condenser have been devised and are advertised by dealers. If a liquid boils at a higher temperature than 170°, there would be danger of fracture if the glass delivery tube were cooled by water. The cooling effect of the surrounding air is, however, sufficient if a long tube be employed. For very volatile liquids, the delivery-tube must be cooled by ice or by a freezing-mixture (pounded ice and salt, or ice and concentrated hydrochloric acid are convenient for moderately low temperatures). In this case a spiral, or "worm," tube should be used (Fig. 10). Condensation of moisture in the receiver is prevented by the drying tube, A.
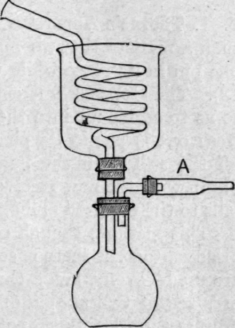
Fig. 10. Condenser for volatile liquids.
Modifications Of The Receiver
If a liquid boils at a very high temperature, or if it suffers decomposition at its ordinary boiling point, it may be necessary to distil it under reduced pressure. For cases of simple distillation the apparatus shown in Fig. 11 may be employed, but if the distillate is to be collected in separate portions, the removal of the receiver would necessitate admission of air into the apparatus and a fresh exhaustion after each change. In order to introduce successive fractions into the still without disturbing the vacuum, Noyes and Skinner fuse a separatory funnel and the still-head to the neck of a Claissen bulb. [The large globe in Fig. 11 serves to keep the pressure steady and to prevent oscillation of the mercury in the gauge.] Various methods have been devised to allow of the receivers being changed without altering the pressure, of which the following may be mentioned.

Fig. 11. - Simple apparatus for distillation under reduced pressure.
1. Thorne's Apparatus. - A series of stopcocks may be arranged in such a manner that air may be admitted into the receiver and a fresh one put in its place while the distillation bulb remains exhausted (Fig. 12). The stopcock b is closed, and the three-way stopcock c is turned so as to admit air into the receiver, which is then disconnected and a fresh one is put in its place. The stopcock a is then closed to shut off the still from the pump, and c is turned so as to connect the pump with the new receiver, which is then exhausted until the pressure falls to the required amount, when a and b are again opened.
This method, though comparatively simple, is attended by several disadvantages; there is some risk of leakage when so many stopcocks are used - even when a three-way stopcock is employed (as in Fig. 12) in place of two simple ones - and this is especially the case because ordinary lubricants cannot as a rule be used for b, through which the condensed liquid flows. Moreover, the changing of the receiver, the manipulation of the stopcocks, and the exhaustion of the fresh receiver take up some time, during which the progress of the distillation cannot be closely watched.
1 Noyes and Skinner, "An Efficient Apparatus for Fractional Distillation under Diminished Pressure," J. Amer, Chem. Soc, 1917, 39, 2718.
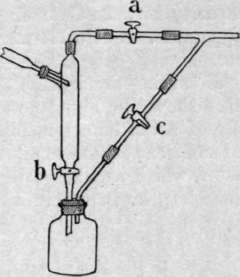
Fig. 12. - Thorne's apparatus for distillation under reduced pressure.
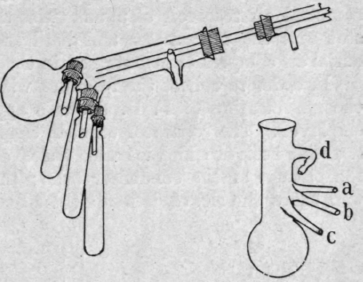
Fig. 13. - Bredt's apparatus for distillation under reduced pressure.
2. Bredt's Apparatus. - To the end of the delivery tube from the still-head is attached a round-bottomed flask with a long neck to which are sealed three narrow tubes a, b, and c, approximately at right angles to it (Fig. 13), and a fourth tube d, which serves to admit air when the distillation is completed. The bulb of the flask serves as one receiver, and each of the three narrow tubes is connected with a cylindrical vessel by means of a perforated cork. The long-necked flask is first placed with the three receivers in an inverted position, so that the first fraction collects in the flask; when a change is to be made, the neck of the flask is rotated until the drops of distillate fall into one of the cylindrical receivers and each of these in turn can be brought vertically below the end of the delivery tube.
3. Bruhl's Apparatus. - A number of test-tubes are placed in a circular stand which may be rotated within an exhausted vessel (Fig. 14) so that any one of the tubes may easily be brought under the end of the delivery tube. This arrangement is convenient, as the change of receiver can be effected with the greatest ease and rapidity.
4. Wade and Merriman's Apparatus for Distillation under Constant Low or High Pressure. - To keep the pressure constant a pressure regulator or "manostat" was devised on a principle similar to that of a thermostat. The air inlet passage is automatically left uncovered by the mercury of a manometer when the pressure falls below the limit to which the manostat is set. For the construction of the instrument and the method of using it, the original c paper1 should be consulted. -In employing the instrument for fractional distillation an air reservoir of about 10 litres capacity is placed between the manostat and the vacuum pump or air compressor and a second air reservoir of about twice the capacity between the mano-stat and the manometer and distillation apparatus. A suitable mercurial air compressor is described in the paper.
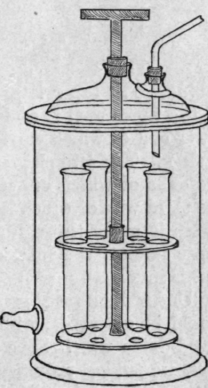
Fig. 14. - Bruhl's apparatus for distillation under reduced pressure.
In order to collect the fractions without disturbing the distillation the modification of F. D. Brown's apparatus shown in Fig. 15 was employed. The separator consists essentially of a tap-funnel B, into the wide neck of which is sealed a sleeve c, of sufficient calibre to allow the passage of the condenser tube ; the latter, which should project well below the sleeve, is made tight by a short length of stout rubber tubing. The tap p is preferably of large bore, 3 or 4 mm. The stem of the tap-funnel is sealed to the upper end of a second sleeve l, the lower end of which carries the rubber stopper of the receiving tube or flask R. The two sleeves are respectively furnished with t tubes tt', the ends of which are sealed to opposite ways of a three-way tap H, the third way of which is open to the air. Connection with the vacuum pump is made through a branch t" of the upper side tube t'. The tap p cannot, as a rule, be lubricated, but as any leakage is inwards no loss is involved.
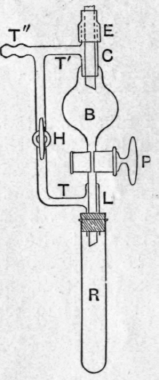
Fig. 15.
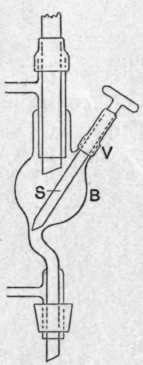
Fig. 16 a.
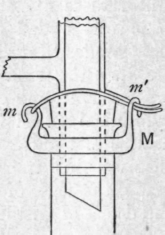
Fig. 16 b.
Under increased pressure a tap is inadmissible, for the leakage would be outwards, whilst the pressure would tend to loosen the tap. For liquids which have no action on rubber a short length of stout pressure tubing provided with a screw clamp forms an excellent substitute. For other liquids the funnel B is provided with an internal stopper s (Fig. 16 a) which can be actuated from outside. This stopper is mounted on a stout glass rod, which passes through a closely-fitting glass sleeve v, to which it is secured by a short length of rubber pressure tubing. This tubing, which is wired to the rod and sleeve, acts as a spring which normally keeps the stopper away from its embouchure in the upper, bent part of the funnel stem. In changing the receiver, the rod is pressed inwards against the elasticity of the rubber tube until it closes the embouchure, and on relieving the pressure by means of the three-way tap, the stopper is held firmly in position by the pressure of the compressed air in the funnel. Any leakage is downwards and internal and the receiver can be changed so quickly that there is no danger of loss. In a distillation under increased pressure all the rubber joints and stoppers must be wired to the respective glass vessels and tubes. A convenient fastening for the receiver is afforded by a stout metal ring m (Fig. 16 6) which fits the test tube fairly closely, and is provided with two stout vertical hooks mm', one of which is radial, the other parallel to the ring ; a loop of fairly stout mild steel wire is passed over the first of these hooks, and the two ends, after traversing the stopper on either side of the sleeve, are sprung under the second, the elasticity of the wire keeping it securely in position.
1 Wade and Merriman, "Apparatus for the Maintenance of Constant Pressures above and below the Atmospheric Pressure. Application to Fractional Distillation," Trans. Chem. Soc, 1911, 99, 984.
Convenient separators have been described by Rosanoff and Easley 1 (see also Rosanoff, Bacon and White),2 and by Hahn.3
Continue to:


