Sublimation. Continued
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Sublimation. Continued
Volatilising Point
The curve ba represents not only the volatilising points of a solid under different pressures but also the vapour pressures of the solid at different temperatures,1 just as the curve AC represents both the boiling points and the vapour pressures of the sub-stance in the liquid state ; the volatilising point of a solid may therefore be defined in precisely the same way as the boiling point of a liquid.
The volatilising point of a solid is the highest temperature attainable by the solid under a given pressure of its own vapour when vaporising with a perfectly free surface and when heat reaches the surface from without.
Dalton's Law Of Partial Pressures
Dalton's law of partial pressures applies to the volatilising point of a solid just as it does to the boiling point of a liquid (p. 25) ; it is the partial pressure of the vapour, not necessarily the total pressure, on which the temperature depends.
Iodine
If a quantity of iodine is heated in a porcelain basin and a cold glass funnel or plate is held over it, the iodine will melt and boil in the basin, but crystals of solid iodine will be deposited on the funnel or plate. The heavy iodine vapour collects in the basin, so that its partial pressure in the atmosphere surrounding the substance soon rises above the triple point pressure, 91 mm., when fusion takes place ; but before reaching the cold glass surface the hot vapour becomes so diluted with air that the partial pressure falls below the triple point pressure, so that when condensation takes place the solid state is at once assumed. In the purification of crude iodine in France by "sublimation," the iodine is heated in earthenware retorts aa (Fig. 84), placed in a sand-bath b, and completely immersed in sand, so that condensation in the upper part of the retorts and necks may be prevented. The short necks of the retorts are connected with large earthenware receivers cc, so that the iodine vapour, on entering the receivers, is rapidly diluted with air. The partial pressure falls below the triple point pressure and direct passage from the gaseous to the solid state takes place. The narrow vertical tubes dd are for the escape of air or of steam if the iodine is moist.
1 Young, Nature, 1881, 24, 239 ; Pettersson, ibid., p. 167 ; Ramsay and Young, Trans. Roy. Soc, 1884, 175, 37 and 461 ; Phil. Mag., 1887 [V], 23, 61 ; Fischer, Wied. Ann., 1886, 28, 400 ; Ferche, ibid., 1891, 44, 265.
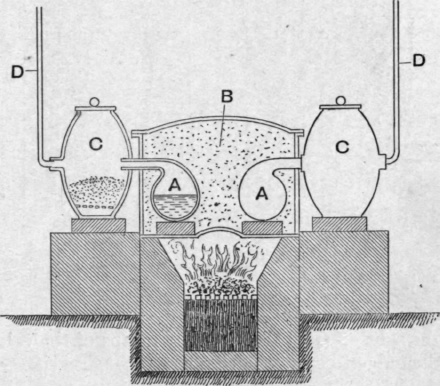
Fig. 84.
The sublimation of the iodine obtained from "caliche" or crude sodium nitrate in Peru and Chili is carried out in a cast-iron retort to which is attached a series of wide cylindrical earthenware receivers or udells connected together so that the condenser as a whole resembles a wide drain pipe. The principle is the same in both cases.
Sulphur
The purification of sulphur by " sublimation," with formation of flowers of sulphur, is carried out in a somewhat similar manner, but the triple point pressure of sulphur is far lower than that of iodine. When sulphur is boiled in an ordinary glass retort the formation of small quantities of flowers of sulphur may always be observed in the upper part of the retort before the air has been expelled, but when, after expulsion of the air, the vapour condenses in the neck of the retort, liquid sulphur is formed.
On the large scale crude sulphur is melted in the pot a (Fig. 85), whence it flows, at a rate regulated by the valve b, through the pipe c into the retort d. It is there boiled and the vapour passes through the wide pipe ee into the large brick chamber F. The sulphur vapour diffuses into the air so that its partial pressure rapidly falls and, so long as the temperature of the chamber is well below the melting point of sulphur, condensation with formation of flowers of sulphur takes place. If the temperature of the chamber is allowed to rise above about 112° liquid sulphur is condensed and this is allowed to flow, as required, through a hole in the plate g and is collected in the pot h. The chamber can be shut off from the retort by means of the valve J, and a workman is then able to enter the chamber through a side door.
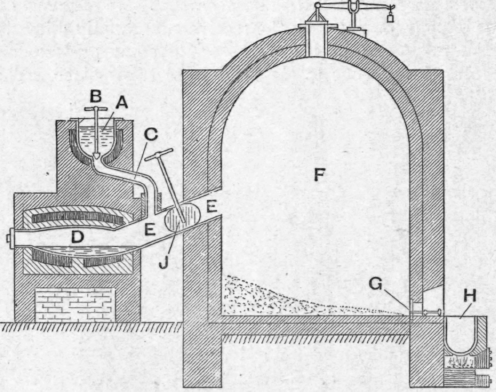
Fig. 85.
Anthracene
In the purification of crude anthracene by passing superheated steam over the melted substance and condensing the vapour in a chamber by jets of cold water, the steam acts as an indifferent gas, and the partial pressure of the anthracene vapour is probably below the triple point pressure when condensation takes place,
Arsenious Oxide
The triple point pressures of arsenic and of arsenious oxide are higher than the atmospheric pressure, and when these substances are heated and their vapours are condensed true sublimation takes place. The method adopted in Silesia in the case of arsenious oxide is illustrated by Fig. 86. The crude arsenious oxide, obtained by roasting arsenical pyrites or other arsenical ores, is placed in the iron pots aa, which are heated each by its own furnace bb. Above the pots are iron cylinders cc covered with caps dd. The arsenious oxide which escapes condensation in the cylinders passes through the pipes ee into the chamber or chambers f. The sublimed oxide is obtained either as a hard vitreous cake or as a loose friable mass according to the temperature at which the operation is conducted.
In Wales the vitreous "white arsenic " or arsenic glass is obtained by causing the sublimation to take place under slightly increased pressure. The crude oxide is heated in a cast-iron pan over which is placed a bell-shaped cover with a hole at the top. After the introduction of the crude oxide, the hole is closed by a plug so that the pressure rises to some extent when the pan is heated. The white arsenic condenses on the cover as a transparent glass.
In the preparation of arsenious oxide by roasting arsenical ores, or when the sublimation takes place in a furnace through which the hot gases pass as well as the vapour of the oxide, the light sublimate is very liable to be carried away, and it is usual to conduct the gases through very long flues or through a series of condensing chambers. Electrical methods of causing the deposition of fine particles are now, however, employed for many purposes.Ammonium Chloride. - When ammonium chloride is heated it does not melt but undergoes almost complete dissociation into ammonia and hydrogen chloride, these substances recombining when the temperature falls. The purification of ammonium chloride by sublimation differs, therefore, from that of arsenious oxide in so far that there is dissociation and recombination in place of vaporisation and condensation, but the methods employed in practice are very similar. The crude ammonium chloride is heated in large cast-iron pots a (Fig. 87), and the sublimate forms on flat or concave iron covers b, which rest on the pots.
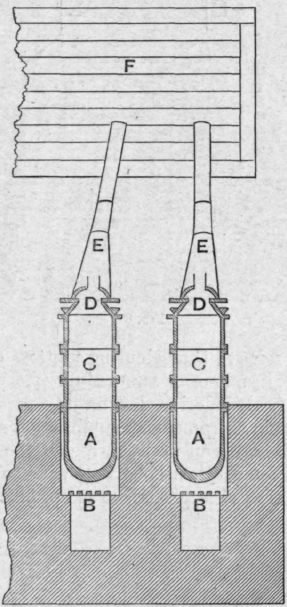
Fig. 86.
Smaller earthenware or glass vessels are also used for the sublimation of ammonium chloride, and the crude chloride is sometimes mixed with animal charcoal to remove the colouring matters derived from tar, or with calcium superphosphate or ammonium phosphate to prevent the sublimation of any ferric chloride that may be present.
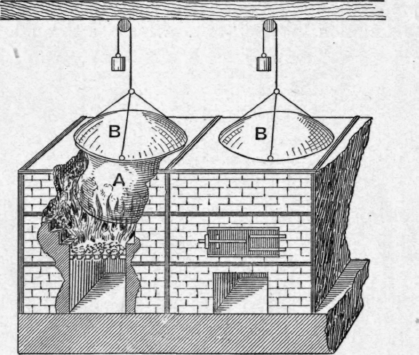
Fig. 87.
It is obvious that in the process of sublimation nothing in the nature of an improved still-head can be used, and if the vapour is conveyed through tubes or pipes, they must be very wide to avoid blocking.
For the sublimation of naphthalene see p. 389.
Continue to:


