The Boiling Point Of A Pure Liquid. Part 2
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
The Boiling Point Of A Pure Liquid. Part 2
Effect Of Impurities On Boiling Point
If there is a rapid rise at first and the temperature afterwards remains steady or nearly so, it may be assumed that there is a much more volatile liquid present. If the temperature is steady at first but rises rapidly when the distillation is nearly complete, the conclusion may be drawn that a much less volatile liquid is present. In either case the steady temperature will approximate very closely to the true boiling point, but it would be more satisfactory, especially in the second case, to collect the best portion of the distillate separately, and to redistil it and read the boiling point again. If, on the other hand, there is a fairly steady rise of temperature throughout, the presence of one or more substances, not very different in volatility from the pure liquid itself, is probable, and it is impossible to ascertain the boiling point of the pure liquid without carrying out a fractional distillation to remove the impurities.
1 Ramsay and Young, " On a New Method of Determining the Vapour Pressures of Solids and Liquids," Trans. Chem. Soc, 1885, 47, 42.
2 J. Soc. Chem. Ind., 1918, 37, 38 T.
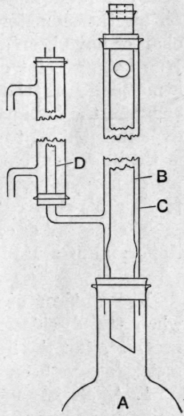
Fig. 18.
Reduction Of Boiling Point To Normal Pressure
Assuming that the liquid is pure, that the thermometer has been compared with an air thermometer, and that the precautions mentioned in the last chapter have been attended to, the corrected temperature will give the true boiling point of the liquid under a pressure equal to that of the atmosphere at the time. It is frequently necessary, however, to compare the boiling point of the liquid with that observed by another experimenter, or to compare it with that of some other liquid, and it is therefore convenient to ascertain what the boiling point would be under normal pressure and, in stating the result for future reference, to give this reduced boiling point.
The method of correcting the boiling point of a liquid from observed to normal pressure is given on p. 13.
Prevention of "Bumping." - It frequently happens, especially when the distillation has to be carried out under greatly reduced pressure, that the liquid is liable to boil with bumping (p. 22), and two methods have been proposed to prevent this.
1. As already mentioned, small tetrahedra of silver or platinum, or fragments of porous material such as unglazed porcelain or pumice-stone, may be added to facilitate the formation of bubbles, or
2. A slow current of air may be admitted through a capillary tube which passes nearly to the bottom of the vessel (Fig. 6, p. 7).
The latter method is an excellent one for preventing the bumping, but it must be remembered that an error is caused by the presence of the air which is introduced. The true boiling point of the liquid and the condensing point of the vapour depend on the pressure of the vapour itself and not necessarily on the total gaseous pressure to which the liquid is exposed. If there is air mixed with the vapour, the total pressure remains unaltered but the partial pressure of the vapour is diminished, and the observed temperature is lower than the boiling point under the read pressure. That this is so may be easily proved by altering the rate at which air enters through the capillary tube ; the more rapid the introduction of air the lower will be the observed temperature, and unless the amount of air is exceedingly small the observed boiling point will be sensibly too low.
Law Of Partial Pressures
Dalton's law of partial pressures is, in fact, applicable to the boiling point of a liquid, as indeed is evident from the fact that in determining a boiling point by the statical method, if air is mixed with the vapour, it is the partial pressure of the vapour and not the total pressure that must be taken into account (p. 21). Many experiments might also be described to prove the truth of this statement, but it will be sufficient to mention the following :Experimental Proofs. - Water, when distilled in the ordinary manner under a pressure of 15 mm., boils at about 18° ; but Schrotter1 observed so long ago as 1853 that when some water was placed in a shallow clock glass, supported on a short tripod on a second clock glass containing strong sulphuric acid, the whole being placed under a bell-jar which could be exhausted by an air-pump, the temperature fell to - 3° when the pressure was reduced to 15 mm. Here the aqueous vapour was rapidly absorbed by the strong sulphuric acid, so that, when the total pressure was 15 mm., the partial pressure of the vapour in contact with the water must have been only about 4 mm. The "boiling point" of the water therefore fell below the freezing point; but in these circumstances bubbles could not be formed (except possibly air bubbles), for the vapour pressure of the water would be far lower than the total gaseous pressure.
More rapid diffusion and removal of vapour, as well as freer evaporation, was effected by suspending in the bell-jar a thermometer, the bulb of which was covered with a piece of sponge soaked in water ; in this case, under a pressure of 40 mm., at which the boiling point of water under ordinary conditions is 34°, the temperature actually fell to -10°. Here the partial pressure of the vapour must have been only a very small fraction of the total pressure.
The following experiment2 affords a still more striking proof of the correctness of the statement that the "boiling point " does not necessarily depend on the atmospheric pressure. A copper air bath, through which a current of air could pass freely, was heated to 205°, and a thermometer, the bulb of which was covered with cotton-wool moistened with boiling water, was suspended in the bath through an opening in the top. The pressure of the atmosphere was 748 mm., and the water on the cotton-wool was in a strongly heated chamber, yet its temperature, instead of remaining at nearly 100°, fell rapidly to 66°, and remained nearly constant at this point. When, however, the current of air through the bath was retarded by closing the grating in the side, the temperature of the water rose to about 80°, and when steam was introduced into the bath so as to replace the air as completely as possible by aqueous vapour, the temperature of the water rose to 99°, though that of the bath had fallen slightly. Lastly, on allowing some of the steam to escape, the temperature of the water fell again to 80°.
In this experiment, it is clear that the temperature of the water did not depend on that of the air bath or on the atmospheric pressure, both of which remained nearly constant. By limiting the supply of air and by introducing steam, nothing was altered but the relative pressures of the aqueous vapour and of the air surrounding the water. The greater the partial pressure of the vapour, the higher was the temperature reached by the water, and when the air was almost completely replaced by aqueous vapour, the temperature rose very nearly to the ordinary boiling point of water.
1 "On the Freezing of Water in Rarefied Air,"Liebig's Annalen, 1853, 88, 188. 2 Young, " Sublimation," Thorpe's Dictionary of Applied Chemistry.
Continue to:


