Sulphur Black T From Dinitrochlorbenzene
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Sulphur Black T From Dinitrochlorbenzene
Reaction:
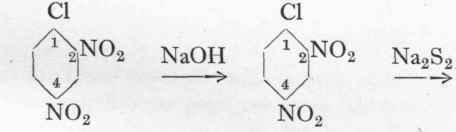
Dye of unknown constitution.
70 Gms. dinitrochlorbenzene are heated to 900 with stirring in a glass or iron vessel with 120 c.cs. of water, and to it is added during 2 hours 108 gms. of 35 % caustic soda solution, so that the reaction never becomes strongly alkaline. Heating is continued until a test portion dissolves to a clear solution in water, a further quantity of caustic lye being added if necessary. The suspension of the sodium salt of dinitrophenol is cooled to 45°, and to it is added a solution of 50 gms. sulphur and 125 gms. crystalline sodium sulphide in 125 gms. water. The temperature is then raised to 6o°, the total volume being 600 c.cs. The mixture is next heated cautiously to 8o° on the water-bath, and then, in the course of 2 1/2 hours, to 105o, on an oil-bath. The mass is now boiled under a reflux for 30 hours without stirring, and is then diluted with 600 c.cs. water. After cooling down to 6o° air is blown through the liquid until the dye is completely precipitated, when it is filtered off and dried at 700. The yield is about 70 gms. Cotton is dyed at the boil, using four times the weight of crystalline sodium sulphide calculated on the weight of dye.
120 c.cs. Water. 70 gms. Dinitrochlorbenzene 108 gms.
35% NaOH.
Notes on Works Technique and Practice. - Sulphur Black T is the most important Sulphur Black on the market, and is superior to other brands as regards fastness to washing and light. Charges of 500-1500 kilos, of dinitrochlorbenzene are made use of in the works, the melt-pots having capacities up to 12,000 litres and the oxidizing vessels up to 30,000 litres. With such large charges it is unnecessary to heat externally, as the heat of the reaction suffices. The pots are made of cast-iron, and are rapidly corroded. From the mother-liquor sodium thiosulphate may be obtained which is used in photography and in the textile industries; a certain portion is also used in the dye-works for the production of Methylene Blue. The price of Sulphur Black T was formerly about 80-90 centimes per kilo, for a 35 % product, so that only those colour factories can make it successfully which are careful to use up all by-products. Further, those colour factories which do not manufacture chlorbenzene and dinitro-benzene are practically out of the running.
50 gms. S. 125 gms. H2O.
125 gms. cryst.
Na2S.
Auramine 00
(according to Traugott Sandmeyer).1
Reaction:
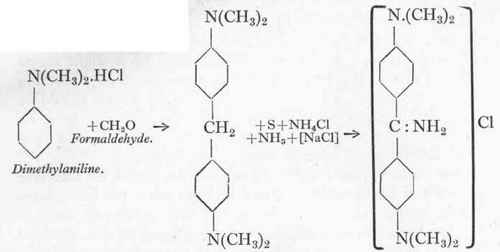
Tetramethyl-diamino-diphenylmethane.
Auramine OO.
(a) Tetramethyl-diamino-diphenylmethane.
242 Gms. of pure dimethyl aniline are mixed with 140 c.cs. of water and 260 gms. of hydrochloric acid (30 %), and to it is added at 300, 60 gms. of 40 % formaldehyde, the strength of which has been accurately determined beforehand by titration. The ratio of formaldehyde to dimethylaniline is 1:1:2 molecules.
The mixture is heated up to 850, with occasional stirring, for 5 hours, and the base precipitated with about 120 gms. of soda dissolved in a little water. The product is filtered off at 200 and is thoroughly washed with water. As the melting point is 900, the drying temperature should not exceed 6o°. Yield about 255 gms., i.e. practically quantitative.
242 gms. Pure dimethyl-aniline.
260 gms. 30% Hc1. 140 gms. H2O. 60 gms.
40 % Ch2o.
About 120 gms. Na2Co3.
(b) Auramine 00.
127 Gms. diamino base, 32 gms. sulphur, 70 gms. ammonium chloride, and 1000 gms. common salt are heated up to 110° in a stirring-pot similar to that shown on Plate XIV., Fig. 36. It is essential that all the substances should be finely divided and should contain no water. The temperature is raised to 130o during 2 hours,2
127 gms.
base.
32 gms. S.
70 gms.
Nh4c1.
1000 gms.
NaCl.
1 In the patent literature Auramine is connected with the meaningless name of Feer. The true discoverer was Sandmeyer, whose name was lost sight of owing to the heated controversy as to the ownership of the colour.
2 Oil-bath about 25° higher.
a rapid stream of dry ammonia being passed into the apparatus from a cylinder. Any moisture is carried away by the gas, and at about 140o a vigorous evolution of hydrogen sulphide begins, which lasts from 5-7 hours, according to the speed of the stream of ammonia. The temperature is raised to 145o during 5 hours, the stirring being continued, and the hydrogen sulphide absorbed in concentrated caustic soda lye. It is also advisable to keep up a slight excess pressure of about 1/5 atms., measured by a manometer, by throttling down the exit tube. The speed of the ammonia stream should be about 5 bubbles per second. Before use the ammonia must be passed through a wash bottle containing 50 % potash, and then through two towers filled with lumps of caustic soda.1
When the evolution of hydrogen sulphide has ceased, the autoclave is opened and the brownish-yellow powdery mass is put into a large porcelain basin. The powder is covered with 3 litres of water to dissolve out the salt, after which the dye is filtered off and dissolved in 1 1/2 litres of water at 6o°. The temperature must not exceed this, as Auramine readily decomposes. The solution is filtered from the residue, consisting of a little sulphur and some Michler's ketone, and the filtrate is mixed with a litre of the salt solution previously obtained, the Auramine coming down in beautiful, glistening, golden leaflets. The yield of pure dry colour may reach 175 gms. It dyes cotton mordanted with tannin and tartar emetic a pure yellow.
Notes on Works Technique and Practice. - Auramine is the most important basic yellow dye, and is much valued owing to its extremely pure shade. The manufacture is carried out in oil-jacketed boilers which must be very accurately heated, as the slightest variation diminishes the yield. Use is also made of Frederking autoclaves, which can be very carefully regulated. Plate VIII., Fig. 24, shows such a flat Auramine pot with modern steam heating. The purity of the salt used has a very considerable influence; traces of the chlorides of calcium or magnesium, which are present in all ordinary "salt, have an injurious effect. The best salt is Galician rock-salt, which is almost chemically pure. The ammonia is dried in small towers filled with caustic soda. Only sufficient ammonia is added to give an excess pressure of half an atmosphere, and the gas is then circulated over the salt mixture by means of a pump. The hydrogen sulphide is absorbed and is used for reductions in the form of sodium sulphide. When the operation is properly conducted the yield may be up to 132 %, i.e. 132 kilos. of pure 100 % Auramine may be obtained from 100 kilos, of tetramethyl-diamino-diphenylmethane. It is difficult to estimate the yield as very few people are able to determine exactly the precise strength of dyeings on tannined cotton. For this reason it is usual to estimate this dye by titrating with titanous chloride of known strength until a weighed portion is rendered colourless, rather than by dyeing trials.
+Nh3.
3 litres H2O.
1 Ammonia cannot, of course, be dried With calcium chloride, as all amines combine with it.
In addition to Auramine OO, Auramine G is also prepared from monomethyl-o-toluidine; it is purer and greener than the 00 brand. The product from diethylaniline is not made as it comes out in such a resinous form on salting out that it is impossible to work it up.
Auramine is used on a very large scale for dyeing cotton, and more especially paper. The Swedish match factories alone use about eight waggon-loads a year for dyeing yellow match-boxes.
Continue to:


