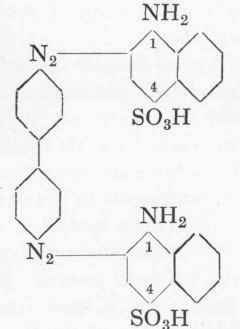
The Intermediate Compound Of Benzidine With Salicylic Acid. Part 4
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
The Intermediate Compound Of Benzidine With Salicylic Acid. Part 4
11 gms. pure m-Phenylene diamine.
10 gms.
Na2C03.
120 gms.
NaCl.
20 c.cs. conc.
Hc1.
Congo Red.
18.6 Gms. of commercial benzidine are diazotized as described on p. no, and this solution mixed with 50 gms. 100 % naphthionate and 50 gms. sodium acetate dissolved in 200 c.cs. water. The temperature is kept for an hour at 50, and is then raised slowly to 200, and kept there for 5 hours. It is then raised to 300, and stirred for 24 hours at this temperature, which is increased again on the third day to 550. When the coupling has proceeded for two and a half days, the mixture is boiled up and treated with 40 gms. of calcined magnesia, which precipitates out the very sparingly soluble magnesium salt of Congo Red, which is filtered off and thoroughly washed. In this manner the impurities are completely removed. The washed magnesium salt is pasted up with 500 c.cs. of boiling water, and is then decomposed with 15 gms. of sodium carbonate, magnesia being precipitated as carbonate and the dye going into solution as the sodium salt. The hot solution is filtered, the magnesium carbonate washed with water, and the Congo Red precipitated from the filtrate by means of 15 (volume) per cent. of common salt. The colour comes out as a bright red precipitate, and after drying gives a yield of about 70 gms. Formula:
18.6 gms. Benzidine, tetrazotized.
50 gms. Naphthionate.
50 gms. Sodium acetate.
40 gms. MgO.
500 c.cs. H2O. 15 gms, Na2Co1.
Notes on Works Technique and Practice. - In spite of its great sensitiveness to acids, Congo Red, the first benzidine colour to be made, is still largely used, as it is one of the most beautiful of the direct cotton colours. It is only prepared by two or three factories at the present time, as its price no longer allows any profit. The commercial product containing about 60 % of salt costs only about 70 centimes per kilo.
On the large scale, the coupling is often carried out rather differently from the laboratory method. The coupling may be speeded up very considerably by mixing the naphthionate solution with the tetrazo-benzidine solution at 85°, very good stirrirg being of course essential. Only small charges can be worked up by this method, but, on the other hand, it is possible to effect 8-10 couplings per diem. The excess naphthionate is frequently recovered.
Besides Congo Red, Benzopurpurin is of great importance, and is prepared from o-tolidine and naphthionate. In this case it is not possible to carry out the coupling hot, as the tetrazo compound of tolidine is too easily decomposed. This colour is somewhat less sensitive to acids than Congo Red and, like the latter, is largely used in the Orient. It would appear that in the non-industrial countries bordering on the Mediterranean Sea, where the atmosphere is free from sulphurous and sulphuric acids, such dyeings are faster than they are with us.
Miscellaneous Azo Dyes. Tropseoline or Orange IV
(Azo Yellow) from Sulphanilic acid and Diphenylamine.
The coupling of sulphanilic acid and diphenylamine affords an interesting example of a mineral-acid coupling. In this case it is not possible to work in either neutral or alkaline solution as the diazo-sulphanilic acid is immediately decomposed by sodium carbonate, and in neutral solution it refuses to react, curiously enough. Further, diphenylamine is completely insoluble in water, so that the coupling must be effected in aqueous-alcoholic solution. It is indeed possible under certain conditions, to work without much alcohol, but in this case so much unchanged diphenylamine remains that it becomes difficult to nitrate the dye. Besides Orange IV itself, its nitro derivatives are also valuable. The coupling in alcoholic solution is therefore to be preferred to that in aqueous solution, especially as the yield is better, so that in this way the small loss of alcohol is fully made up for.
As already noted above, diphenylamine has an injurious effect on the nitration, and other impurities which accompany Orange IV possess this property to an even greater extent. If it is intended to convert Tropaeoline into Azo Yellow, it is absolutely necessary that the Tropaeoline acid shall be quite pure, as slight impurities diminish the yield by 30-50 %. If a pure dye is available, then the actual method of nitration becomes comparatively unimportant. This process is of interest, as the nitro compound is obtained via the nitrosamine and nitramine, the Tropaeoline being nitrosated with nitrous acid and then oxidized with very dilute nitric acid. The nitramine is formed intermediately, and is then converted at once into the nitro compound under the influence of mineral acid, exactly as Bamberger's phenyl nitramine is transformed into ortho-nitraniline.
The same relationships are also found in the case of Methylene Green (q.v.).
Owing to its pure shade and satisfactory fastness to light and washing, Tropaeoline is much used for wool. When mixed with certain dyes it increases their strength considerably. This is particularly the case with the much-used Acid Black 4B (a mixture of about 45 % Naphthol Blue Black B and Naphthylamine Black D, together with 5 % each of Tropaeoline and Fast Red AV). Attempts to replace Tropaeoline in this case by other yellow colours have shown that the only one having a similar action is Metanil Yellow, which is formed from metanilic acid and diphenylamine. The increase in strength amounts to about 30 %.
For silk, the acid-fast Azo Yellow is used, which goes well on to silk which has been weighted with tin phosphate. For the production of yellow and brown shades fast to water Azo Yellow has become indispensable.
(a) Tropceoline or Orange IV.
Reaction:
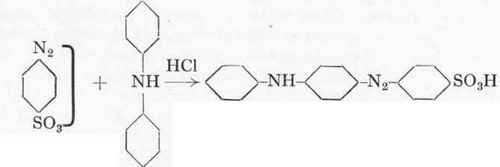
Diazo -sulphanilic acid.
Continue to:


