The Intermediate Compound Of Benzidine With Salicylic Acid. Part 3
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
The Intermediate Compound Of Benzidine With Salicylic Acid. Part 3
Example of the combination, in presence of Mineral Acid, of an Amine which couples readily, with an Amino-naphthol sulphonic acid which couples with difficulty:
Direct Deep Black EW (Bayer).
Formula:

{Benzidine.)
In the case of Diamine Green B we have become acquainted with a mineral-acid coupling of H-acid with p-nitraniline, and have seen that these components combine readily to give a monoazo dye in which the azo group is attached in the ortho position to the amino group. Benzidine couples far less readily, and it is necessary to neutralize continuously the mineral acid which is set free. Contrary to the data given in the patent literature, it is not possible to carry out this reaction in acetic acid solution, since H-acid in presence of sodium acetate at once couples in the ortho position to the hydroxyl. This fact has given rise to many patent actions, which, however, have all been decided in favour of the patentee of the mineral-acid coupling process.
(a) The intermediate compound:
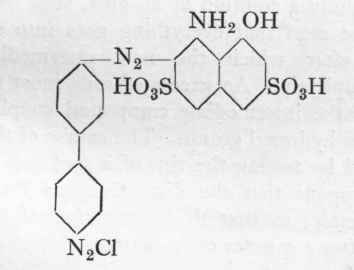
19.2 Gms. of 100 % benzidine are diazotized as described on p. no, and the temperature reduced to 10-12o. To this tetrazo solution is added during the course of an hour the filtered solution of 34.1 gms. H-acid dissolved in 5.5 gms. soda and 300 c.cs. of water. The H-acid solution should react distinctly acid to the litmus. Stirring is continued at 12o for 3 hours, after which a solution of 5.5 gms. soda in 60 c.cs. water is run in very cautiously during 2 hours, care being taken that the mineral acid reaction never disappears for a moment. After a further 3 hours at 12o sufficient dilute sodium carbonate solution is added, if necessary, to give a faint but distinct reaction with Congo paper, the mixture then being allowed to stand all night in a cool place. The reaction for benzidine (with H-acid solution), and also that for H-acid (with diazotized nitraniline), by spotting on filter-paper, will have disappeared completely after 12 hours. The intermediate compound separates out as a powdery precipitate.
(b) The intermediate compound:
19.2 gms. 100% Benzidine, tetrazotized.
34.1 gms. H-acid.
5.5 gms. Na2Co3.
300 c.cs. H2O.
5.5 gms. Na2Co3 in 60 c.cs. H2O

8.8 Gms. of pure aniline are diazotized as described on p. 108, and the diazonium solution is added to the first intermediate at 50, some ice being added if necessary. The mixture is well stirred, and to it is added very rapidly a solution of 26 gms. soda in 120 gms. cold water. It will be seen that everything goes into solution almost instantaneously, after which the new intermediate compound separates out completely. An excess of soda must not be used, or else a portion of the intermediate compound couples up with the H-acid next to the hydroxyl group. The course of the reaction may easily be followed by testing the rim of a spot-test on filter-paper. It sometimes happens that the diazo benzene reaction does not disappear completely, so that the preparation of the final dye is proceeded with after a quarter of an hour.
8.8 gms pure Aniline, diazotized.
26 gms. Na2Co3 in 120 gms. H2O.

Fig. 29. - Oil-jacketed autoclave with duplex stirring gear for stiff melts (scale 1 :25).
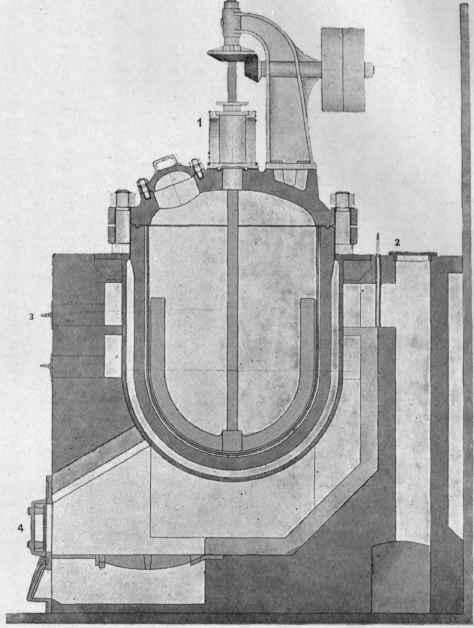
Fig. 30. - Section through an autoclave (scale 1:40). 1. Water-cooled stuffing-box. 2. Flue with cover and damper. 3. Flue doors for cleaning. J4. Furnace doors.
Plate XI.
To the second intermediate compound are added 11 gms. of purest m-phenylene diamine dissolved in a little water, which couples up rapidly with the diazo compound, a portion of the colouring matter formed always going into solution. After 1 hour at 14o the product is heated up cautiously to 50o, and 10 gms. sodium carbonate are added. 120 Gms. salt are then sprinkled in, after which the mixture is acidified with about 20 c.cs. concentrated hydrochloric acid, stirring being maintained until the dye is completely precipitated. It is insoluble in a 10 % solution of common salt and bicarbonate at 500, so long as it has not previously been boiled up. The product filters very readily and, after pressing, is dried at 100°. The yield is about 100 gms. of strong dye. To ensure that the dye goes properly on to cotton it must be mixed with 6 % of its weight of sodium carbonate.
By the use of m-toluylene diamine instead of phenylene diamine, Deep Black V is obtained, which possesses a somewhat more reddish shade. In this case also it is necessary to add a little soda after warming up, in order to obtain an easily filterable product.
Notes on Works Practice. - The cotton black just described is the most largely used direct black made in the dye industry. It is used for dyeing all manner of organic materials such as cotton, wool-mixtures, leather, etc. It is prepared in very large azo plants, only the very purest intermediates being employed. By using m-phenylene diamine which has been recrystallized from water, the highest yields of colour are obtained, the products dyeing pure black ever when the dye-bath is approaching exhaustion. It is included in the so-called "Black Convention" (Schwarz Konvention) concluded between the big colour factories in order to keep the price up to a reasonable level. The price of the most highly concentrated product which was often only diluted with 3 % of salt and 5 % of soda, was formerly about 3 francs, and even less, per kilo.
Continue to:


