The Intermediate Compound Of Benzidine With Salicylic Acid. Part 2
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
The Intermediate Compound Of Benzidine With Salicylic Acid. Part 2
The colouring matters from both phenylene and toluylene diamines are used in large quantities for the production of mixed shades.
Diamine Green B (Cassella).
Formula:
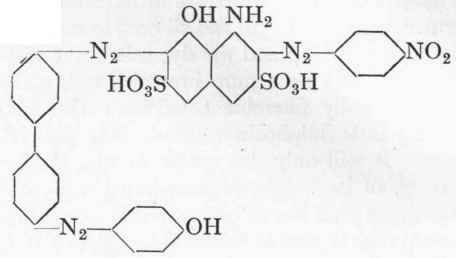
14.5 Gms. pure p-nitraniline are diazotized as described on p. 109, and to the clear, ice-cold diazonium solution is added 34.1 gms. 100 % H-acid dissolved in 200 c.cs. of cold water and 5.5 gms. sodium carbonate. The addition will occupy about three-quarters of an hour, and care must be taken by means of good mechanical stirring that no lumps are formed. The H-acid combines with the nitraniline in 4-5 hours, setting free an equivalent portion of hydrochloric acid. The mixture is now allowed to stand for at least 12 hours, and next day, after heating up to 500, 20 gms. of 30 % caustic soda solution are added and 40 gms. soda. The monoazo dye of the formula:
14.5 gms.
Diazotized p-nitraniline
34.1 gms. 100 % H-acid.
200 c.cs. H2O.
5 gms. Na2Co3.
20 gms.
30 %
NaOH.
40 gms. Na2Co3.

goes into solution with a fine blue colour, and is then salted out by means of 200 gms. common salt. After a few hours, the glistening sodium salt separates out in an easily filterable form, after which it is filtered off and pressed. The mother-liquor has a strong blue colour, but does not yield any further quantity of usable dye on saturating with common salt and is therefore thrown away.
If instead of separating out the dye from p-nitraniline this is coupled with a calculated quantity of diazotized aniline at 50, the important Naphthol Blue-Black B (C.) is obtained, having the structure:
200 gms. NaCl.
40 gms. Na2Co3.
9.3 gms.
Diazotized aniline.
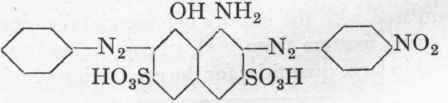
In this case it is unnecessary to isolate the monoazo dye, but an excess of diazotized aniline has a deleterious influence. By salting out at 900 with 15 % of common salt, the Naphthol Blue-Black B is obtained in a fine bronzed form. In passing, it may be mentioned that by reducing Naphthol Blue-Black B with sodium sulphide at 25°, a valuable dark green azo dye is obtained, Azo Dark Green, having the following formula:
50 gms. Na2S.9H20 25o C
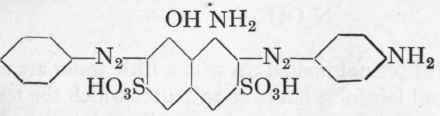
The dye is precipitated out at 50o after standing for 3 hours, with 15 % of common salt and a little sulphuric acid. It is sparingly soluble in bicarbonate; the mother-liquor is deeply coloured.
The following general rule may be noted: Dyes from para-nitraniline may be reduced almost quantitatively to the corresponding p-phenylene diamine azo colours by means of the calculated quantity of sodium sulphide:
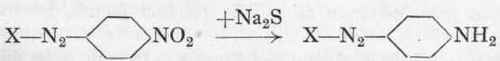
In this way new amino-azo dyes are obtained which may be again diazotized and coupled up with other components. The same amino-azo dyes may also be obtained by hydrolysis of the corresponding p-amino-acetanilide colours.1
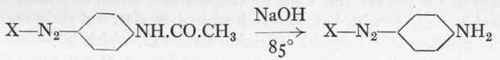
The sodium salt is dissolved in 500 c.cs. water containing 40 gms. soda at 8o°, and is then allowed to cool down to 200 with continuous stirring. Sufficient ice is then added to bring the temperature down to 40; a portion of the dye separates out again in a fine state of division. To this suspension is added slowly a solution of tetrazo-benzidine prepared as described on p. no, the solution being added until a drop on filter paper gives a weak but distinct blue coloration on touching the rim with alkaline H-acid solution. At first the discoloration always fades away again, so that a further quantity of the tetrazo solution must be added. Altogether about 18.6 gms.
500 C.CS. H2O.
40 gms. Na2Co3.
About 18.6 gms. tetrazotized benzidine.
1 Formyl- and oxalyl-p-phenylene diamines may also be utilized.
benzidine must be used, the formation of the intermediate compound taking half an hour.
Formula of the intermediate compound:

12 Gms. of phenol melted up with a little water are added to this compound and left for 3 hours at 10°, after which the temperature is slowly raised to 30o, and the mixture allowed to stand over-night. It is then heated up to 6o°, and sufficient 30 % caustic soda-lye added to bring everything into solution (about 40 gms.).1 150 Gms. common salt are now added, and dilute sulphuric acid dropped in cautiously until the colouring matter is precipitated (test by spotting on filter paper); the latter is filtered off, pressed, and dried at 900. Yield about no gms. strong dye. Instead of using caustic soda for the separation the product may be heated up to 900 and salted out hot with 300 gms. common salt. The product is not, however, so strong.
Notes on Works Technique and Practice. - In spite of its relatively poor fastness to light, Diamine Green B is one of the most largely used green cotton dyes. It serves for dyeing the cotton layer used for insulating the copper wires for telephone and other cables, and for the production of mixed shades.- If salicylic acid be used in place of the phenol, this must be coupled first of all, as it does not combine readily with benzidine when used as a second component. The product so obtained is Diamine Green G, which is, however, much less used as the formation of this colour does not take place so smoothly and, in consequence, the price is considerably higher. In the works, the heating is always effected by blowing in steam, and these dyes cannot be pressed as they are simply forced through the filter-cloths.
12 gms. Phenol.
About 40 gms. 30% NaOH.
150 gms. NaCl. About 50 gms.
50% H2So4.
300 gms. NaCl.
1 Nitro-azo dyes must not be treated with soda-lye in presence of wood or other reducing substances.
Continue to:


