The Intermediate Compound Of Benzidine With Salicylic Acid
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
The Intermediate Compound Of Benzidine With Salicylic Acid
(o-Tolidine → o-Cresotinic Acid.)1
18.6 Gms. (1/10 mol.) commercial benzidine are tetrazotized as described on p. no. The clear tetrazo solution is poured rapidly into a solution of 15 gms. pure salicylic acid and 40 gms. anhydrous sodium carbonate, and 300 c.cs. water at 50. The orange-yellow can combine with numerous amines and phenols. Certain of the dyes produced are important. On adding to the intermediate product, for example, a solution of r-acid made alkaline with sodium carbonate, Diamine Brown V (Cassella) is formed. It is of interest to note that only 85 % of theory of r-acid is required. If, on the other hand, the intermediate compound is acidified with acetic acid, and is then treated with a solution of r-acid which is still distinctly acid to litmus, the important Diamine Fast Red F is formed in about 12 hours (at 12-28°), which, owing to the presence of the salicylic acid group, affords chrome mordanted dyeings on wool which are fast to milling.
18.6 gms.
Tetrazotized benzidine.
15 gms.
Salicylic acid.
40 gms. Na2Co3.
1 The compound o-tolidine → o-cresotinic acid is largely made, whereas salicylic acid cannot be used, as the dyes from o-tolidine → salicylic acid are very sparingly soluble.
intermediate compound separates out, and the end of the reaction may be recognized by placing a drop on filter-paper and testing the colourless rim of liquid with alkaline H-acid, when no blue coloration should be given. Stirring is continued steadily until the benzidine reaction has quite disappeared, which will take about 1 hour at 12o.
The azo compound, which has the formula:
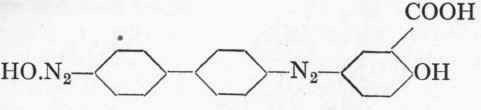
Formula of Diamine Brown V:

Formula of Diamine Fast Red F:

In alkaline couplings the azo group takes up the ortho position to the hydroxyl group, whilst in acetic acid coupling the ortho position to the amino group is attacked.
Dianil Brown 3GN.
Formula:
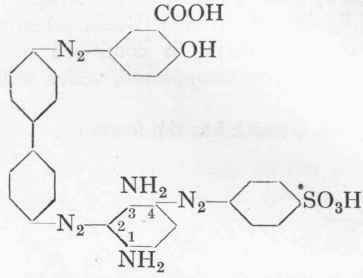
This dye is one of the most frequently used direct azo colours, as it is extraordinarily strong. It is, however, not fast to acids or light. First of all the monoazo dye Sulphochrysoidine G: is prepared in the following manner: 17.3 gms. (1/10 mol.) sul-phanilic acid are diazotized as already described (cf. p. 113), and the suspension of the diazo compound, which must be slightly mineral acid, is allowed to drop slowly into a solution of 10.8 gms. purest m-phenylene diamine. Preferably the diamine is acidified with 5 c.cs. concentrated hydrochloric acid and the solution made up to 10 % diamine. The coupling is followed by means of alkaline H-acid solution "spotted" upon a filter-paper, and the diazo compound is added until a very faint red colour can be detected on the rim. The diamine base disappears completely, but the true azo colour is not yet formed. After standing for 2 hours at 5o, a 10 % soda solution is run in cautiously with continuous stirring, until the mineral acid has been fully neutralized, about 6 gms. soda being required for this purpose. After standing a further 3 hours at 50, 5.5 gms. soda are added during 1 hour, and the whole allowed to stand over-night. Next morning a further 10 gms. of sodium carbonate dissolved in a little water, are added, and the whole again allowed to stand for 3 hours. It is most essential to effect the coupling of the sulphanilic acid and the diamine extremely carefully, or else the final colour will be weak. The sodium salt of the Sulphochrysoidine separates out for the most part as a reddish-brown, beautifully crystalline precipitate. The volume of the whole solution may be about 500 c.cs., but should not be too dilute. This suspension is mixed at 10° with the intermediate compound benzidine → salicylic acid (cf. p. 118), and the mixture stirred steadily for 5 hours. It is then warmed cautiously to 300 and allowed to stand for 12 hours. The liquid is then boiled up and the dye salted out with 200 gms. common salt. It should be a pure brownish red, and should be precipitated in an easily filterable condition; the mother-liquor only contains very little Sulphochrysoidine. The yield of dry colour is about 95 gms. It will only dye cotton evenly, however, if it is mixed with 10 % of its weight of dehydrated sodium carbonate; too little or too much soda has an unfavourable influence, so that we have here a case similar to that of Direct Deep Black EW (cf. p. 118).
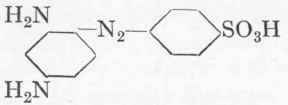
1/10 mol. Diazotized sulphanilic acid.
10.8 gms.
m-Phenylene diamine.
5 c.cs. Hc1.
About 6 gms. Soda.
About 5.5 gms. Soda. 10 gms. Soda.
Notes on Works Practice. - By the use of 1:2:4 toluylene diamine instead of m-phenylene diamine an analogous dye is obtained which is, however, somewhat faster to acids. As in this case the para position to the amino group is occupied, it follows that the formula given above for the m-phenylene diamine colour is correct, i.e. the second azo group is attached between the two amino groups and not in the position para to the Nh2.
If the diamine is first coupled with the benzidine-salicylic acid compound and then with the sulphanilic acid, an isomeric dye is produced of the following formula, which, curiously enough, however, is quite valueless:
1/10 mol. Benzidine-salicylic acid.
200 gms. NaCl.

It is very important to use quite pure diamine, as traces of 0-or p-diamine decompose a large portion of the diazo-sulphanilic acid, and also of the intermediate compound, benzidine-salicylic acid. The solution foams up and the dye becomes weak and muddy. By the use of the purest materials the yield is increased by about 40 %, as compared with the impure commercial diamine solution.
Continue to:


