Oxygen
Description
This section is from the book "The Engineer's And Mechanic's Encyclopaedia", by Luke Hebert. Also available from Amazon: Engineer's And Mechanic's Encyclopaedia.
Oxygen
Oxygen which, uncombined, is known only as a gaseous substance, was discovered by Dr. Priestley, in 1774. It has been called dephlogisticated air, empyreal air, and vital air. The term oxygen was given on the supposition that it was the sole cause of acidity. This substance is highly important in the economy of nature, as it forms about a fifth part of our atmosphere, and is abundantly contained in water, acids, salts, and oxides.
Oxygen gas may be obtained from a variety of sources. The peroxide of manganese, of lead and mercury, also nitre and chlorate of potash, yield large quantities when exposed to a red heat. The substances most commonly employed to procure it, are nitre, peroxide of manganese, and chlorate of potash. if nitrate of potash be exposed to a red heat in a coated glass or earthen retort, or in a gun barrel, a quantity of oxygen gas, (about 1200 cubic inches from a pound of nitre,) will be given off, but this is liable, particularly towards the end of the process, to be contaminated with nitrogen. From the peroxide of manganese, the gas may be obtained either by beating the substance red hot in a
Fig. 4.
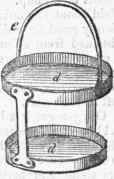
Fig. 5.
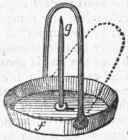
F F gun barrel, or by putting it in the state of a fine powder into a flask, with about an equal weight of concentrated sulphuric acid, and heating the mixture by means of a lamp. In the dry way, one ounce of peroxide of manganese should yield about 128 cubic inches of oxygen. The gas procured in this way is sufficiently good for ordinary purposes, but when required of great purity, it is better obtained from chlorate of potash. For this purpose the salt is to be put into a green glass retort, and heated to redness. It first liquefies, and then on increase of heat, is wholly resolved into pure oxygen gas, which escapes with effervescence, and into a white compound called chloride of potassium, which is left in the retort.
Oxygen gas is a little heavier than atmospheric air. Its specific gravity is 1.111; one hundred cubic inches weighing 34.454 grains. It is sparingly absorbed by water, 100 cubic inches dissolving only 3 or 4 of the gas; but under great pressure it may be made to take up half its bulk. It has neither acid nor alkaline properties, for it does not change the colour of vegetable blues, nor does it evince any tendency to unite with acids or alkalies. It has neither smell nor taste. It refracts light feebly, and is a non-conductor of electricity. It is the most perfect negative electric we possess, always appearing at the positive pole when any compound containing it is submitted to galvanism. It is essential to the support of animal life: an animal will live in it a considerable time longer than in atmospheric air; but its respiration,becomes hurried and laborious before the gas is consumed, and it dies, though another animal of the same kind can sustain life for a certain time in the residuary air. When suddenly compressed, it has been seen to emit light and heat, but this is said to arise from the combustion of the oil with which the tube is lubricated. It has a very powerful attraction for most simple substances, and there is not one of them with which it may not be made to combine.
Any inflammable substance previously kindled and introduced into it, burns rapidly and vividly. If an iron or copper wire be introduced into a bottle of oxygen gas, with a bit of lighted touchwood or charcoal at the end, it will burn with a bright light, and throw out a number of sparks. The bottom of the bottle should be covered with sand, that the sparks may not crack it. If the wire, coiled up in a spiral, like a corkscrew, as it usually is in this experiment, be moved with a jerk at the instant a melted globule is about to fall, so as to throw it against the side of the glass, it will melt its way through in an instant, or if the jerk be less violent, lodge itself in the substance of the glass. If it be performed in a bell glass, set in a plate filled with water, the globules will frequently fuse the vitreous glazing of the plate, and unite with it so as not to be separable without detaching the glaze, though it may have passed through, perhaps, two inches of water.
All substances that are capable of burning in the open air burn with far greater brilliancy in oxygen gas. A piece of wood, on which the least spark of Tight is visible, bursts into flame the moment it is put into a jar of oxygen; lighted charcoal emits beautiful scintillations; and phosphorus burns with so powerful a light, that the eye cannot bear its impression. The act of combining with oxygen is called oxidation, and bodies which have united with it are said to be oxidized. The compounds so formed, are divided into acids and oxides. The former includes those compounds which possess the general properties of acids, and the latter comprehend those which not only want that character, but of which many are highly alkaline, and yield salts by uniting with acids. Oxidation is sometimes produced with great rapidity, and with evolution of heat and light. Ordinary combustion is nothing more than a rapid oxidation, and all inflammable or combustible substances derive their power of burning in the open air from their affinity for oxygen.
On other occasions it takes place slowly, and without any appearance of heat and light, as is exemplified by the rusting of iron when exposed to a moist atmosphere.
Continue to:


